A 40-year-old female is evaluated for uncontrolled hypertension. Home medications include olmesartan and nifedipine. She does not smoke or drink alcohol. BMI is 24 kg/m2. Standardized office BP measurement is 150/85 mm Hg. Ambulatory BP measurement is 140/80 mm Hg. Renal ultrasonography does not suggest renal artery stenosis.
Which of the following is the next best step in management?
A. Obtain computed tomography imaging
B. Obtain 24-hour urine sodium and potassium
C. Obtain albumin-to-creatinine ratio
D. Initiate chlorthalidone
What is (D)
This patient has uncontrolled hypertension. She is already adherent to several lifestyle modifications, including abstinence from tobacco and alcohol. There is no evidence for renal artery stenosis on the basis of the ultrasonography findings. Therefore, there would be no need to obtain CT imaging. Also, there is no need for additional laboratory testing at this time, making answers B and C incorrect. On the basis of the answer choices provided, the correct answer would be to initiate chlorthalidone, answer D, on the basis of evidence of the CLICK trial.
A 24-year-old woman presents to the emergency department after 3 days of fatigue, malaise, tingling in her legs, and restlessness. She also endorses nausea but denies emesis. Her medical history is notable for hospitalization at age 18 years for management of disordered eating, but she reports that this condition resolved before she entered college. She also has a history of persistent hypokalemia that was first identified when she was a teenager. She states she was informed that the hypokalemia relates to a genetic condition that results in excessive renal potassium excretion. Her medications include a potassium supplement of uncertain dose and calcium carbonate, which she takes occasionally for upset stomach. She is a graduate student and is due to present an update on her dissertation soon. She describes herself as highly energetic and often sleeps only 2–3 hours each night. She typically spends several hours each day dancing for exercise and stress reduction.
Her BP while seated is 119/75 mm Hg, heart rate 89/min, respiratory rate 17/min, and body mass index 22 kg/m2. After she stands for 2 minutes, BP is 82/62 mm Hg and heart rate 121/min. She is alert and oriented. She appears anxious. Porcelain crowns are present on her anterior teeth. The heart and lungs are normal. Her abdomen is diffusely tender, but there is no guarding or rebound. Mild pretibial edema is present.
Laboratory data include the following:

Which of the following is the MOST LIKELY explanation for her condition?
A. Milk-alkali syndrome
B. Gitelman syndrome
C. Bartter syndrome
D. Disordered eating
E. Diuretic abuse
What is (D)
The most likely diagnosis for this patient with metabolic alkalosis, hypokalemia, and low urine chloride is vomiting related to disordered eating of the subtype bulimia with binging-purging behavior.
Eating disorders are often concealed by patients who are not forthcoming about their behavior or may not recognize their behavior as pathologic. The diagnosis is particularly challenging in patients such as this one, in whom alternative diagnoses may have been erroneously postulated and then propagated.
The differential diagnosis of metabolic alkalosis includes gastrointestinal hydrogen ion loss, renal hydrogen ion loss, and the addition of exogenous alkali. In this case, the divergence of urine chloride (very low) and urine sodium (not low) suggests vomiting as the primary driver of the derangements. Vomiting results in loss of hydrochloric acid in the emesis, which is accompanied by the addition of bicarbonate ion to blood. The resulting high level of filtered bicarbonate then exceeds the resorptive threshold in the proximal tubule and functions as a non-reabsorbable anion, carrying sodium and potassium into the urine. Urine chloride is low because of concomitant volume contraction and increased reclamation of chloride along the nephron, as well as a reduced filtered load due to the loss of chloride in the emesis. If a steady state is reached and emesis ceases, urine sodium decreases under the influence of aldosterone, but the alkalosis persists until chloride is replete. Of note, hypomagnesemia is commonly identified in patients with disordered eating. Inadequate intake and urinary losses may contribute. Refeeding can exacerbate this problem.
A 22-year-old man with a history of recurrent nephrolithiasis due to cystinuria is seen in clinic for follow-up. He underwent percutaneous nephrolithotomy 9 months ago for a right-sided staghorn calculus. Postoperative imaging showed no residual stone burden. He has passed several small stones since surgery but currently denies visible hematuria, dysuria, or flank pain. He is tolerating potassium citrate for urinary alkalization.
On examination, his BP is 123/69 mm Hg and heart rate is 74/min. He is in no apparent distress. The abdomen is soft and nontender. The remainder of the examination is unremarkable.
Laboratory data are as shown below:

Kidney ultrasound demonstrates bilateral, nonobstructing calculi up to 7 mm in diameter.
In addition to encouraging increased fluid and decreased sodium and protein intake, which ONE of the following is the MOST appropriate treatment for this patient?
A. Tiopronin
B. α-Lipoic acid
C. Indapamide
D. Acetazolamide
E. Tolvaptan
Tiopronin, a thiol-containing medication, should be initiated for resistant cystine stone disease.
Conservative therapy for cystinuria is targeted to increase cystine solubility by maximizing urinary volume and alkalinization. Fluid intake goals may vary depending on the individual but should be prescribed to maintain the urinary cystine concentration below 250 mg/L. Patients are encouraged to drink throughout the day and night to prevent increases in cystine supersaturation overnight. The solubility of cystine increases significantly with a urine pH above 7. Therapy with potassium citrate or potassium bicarbonate is given in divided doses throughout the day to maintain 24-hour urinary pH at 7.5–8.0. Moderate sodium and protein restrictions are recommended to reduce urinary cystine excretion. Lower protein intake also aids urinary alkalinization via decreasing net acid excretion. Thiol-containing medications are indicated when conservative therapy fails to control cystine stone disease. Thiol-containing drugs have sulfhydryl groups that reduce the disulfide bond between the cystine homodimer and form drug-cysteine complexes that are significantly more soluble than cystine.
The cystine-binding drugs tiopronin and d-penicillamine are effective in reducing free urine cystine levels. Both medications carry a risk of severe adverse events, including neutropenia, aplastic anemia, hepatotoxicity, and proteinuria attributed to membranous glomerulopathy. Patients treated with thiol agents should undergo regular monitoring of liver enzymes, complete blood cell counts, and urinary protein excretion. d-Penicillamine is suggested to have a higher incidence of adverse effects.
A 65-year-old man with ESRD due to type 2 diabetes and hypertension presents with a 1-year history of decreased libido and sexual function. He receives HD thrice weekly. His medical history also includes coronary artery disease and prostate cancer currently being managed with watchful waiting. He reports that most times, he is unable to achieve erections, and when he does, they are not hard enough to engage in sexual intercourse. This problem has become an increasing source of friction between him and his partner and significantly affects his quality of life.
Laboratory data are as follows:

Kt/V is 1.4.
Which of the following is the NEXT BEST STEP to manage sexual dysfunction?
A. Sildenafil
B. Citalopram
C. Calcitriol
D. Bromocriptine
E. Testosterone
What is (A)
The next step in treating this patient with ESRD and erectile dysfunction is to prescribe a phosphodiesterase-5 inhibitor such as sildenafil.
Erectile dysfunction (ED) is common in patients with ESRD, affecting 80% and rated as severe in 50%, often adversely affecting quality of life. In this case, the patient has ED that is severe enough to adversely affect his relationship with his partner. Therapy with a phosphodiesterase-5 inhibitor such as sildenafil can improve ED and is usually well-tolerated. Medications in this class should be avoided by patients receiving nitrates. Interactions with other drugs such as α-adrenergic blockers can also be a concern.
Prolactin levels are often elevated in men receiving dialysis, as in this case. The cause of mild hyperprolactinemia is unknown but may be related to elevated PTH levels. Hyperprolactinemia, particularly when extreme, is associated with sexual dysfunction in men with ESRD. Although bromocriptine has been shown to lower prolactin levels to near normal, the effect on sexual function has been inconsistent. Hypotension may also limit the use of bromocriptine. A trial of bromocriptine could be considered if the response to sildenafil is inadequate, but it would not be the first choice.
The patient is a 62-year-old male with ESKD with a past history of type 2 diabetes and hypertension. The patient originally had his lisinopril held while initiating hemodialysis, to allow for appropriate dialysis without hypotension. Because of maintained hypertension, resumption of antihypertensive therapy is warranted.
What therapeutic option is best supported by current data (albeit limited) to manage this patient?
A. Resume lisinopril because of the patient’s history of compliance and history of diabetes
B. Change to carvedilol to manage hypertension and reduce the risk of fatal arrythmia
C. Change to hydralazine for BP control
D. Change to amlodipine for BP control, given the improvement in major adverse cardiovascular events for calcium channel blockers not seen with ACEI
What is (D)
This patient has no compelling indication to select any specific agent
because of comorbidities, and so should make a selection on the basis of BP management along
with cardiac outcome data. Although data are limited, amlodipine has better cardiovascular
outcomes data on the basis of a recent placebo-controlled trial and a retrospective analysis.
Carvedilol does have some data on reducing MACE, but in those with cardiomyopathy, none of
which was noted in this case. Resuming lisinopril without a specific indication would not be
recommended, because of the lack of mortality and cardiovascular benefit noted in studies.
A 29-year-old man presents to the emergency department because of frequent episodes of vomiting and signs of dehydration. The patient reports non-bloody emesis for >48 hours. He has not traveled abroad recently, has no fevers or chills, and has no diarrhea. He is otherwise healthy and does not take any medication on a regular basis. He drinks two to three alcoholic drinks per week and reports to consume marijuana frequently.
His partner, who accompanies the patient, reports that the patient had similar episodes of vomiting during the past 2 years for which he needed medical attention.
The patient has a BMI of 18 kg/m2, a blood pressure of 98/52 mm Hg, a pulse of 104/min, and a respiratory rate of 18, and is afebrile. Clinical examination is unremarkable.
Laboratory investigations reveal a serum potassium level of 2.4 mEq/L, sodium of 135 mEq/L, and serum creatinine of 1.16 mg/dl (102 µmol/L). Venous blood gas analysis reveals a bicarbonate level of 38 mEq/L.
What is the most likely mechanism leading to this patient’s hypokalemia?
A. Loss of potassium via gastric fluid in combination with a low potassium intake
B. The combination of increased sodium bicarbonate delivery to the collecting duct and increased volume depletion–induced aldosterone activity
C. Bulimia nervosa is the underlying problem: the patient consumes large amounts of laxatives and diuretics
D. Potassium is shifted intracellularly because of hyperventilation, explaining hypokalemia
E. The patient is in distress, causing a hyperadrenergic state, which, in turn, causes hypokalemia
What is (B)
Answer B is correct because the answer summarizes the mechanisms leading to hypokalemia in prolonged, severe cases of vomiting. Answer A is incorrect because the potassium content in gastric fluid is low. Answer C is incorrect because there is no suggestion in the clinical history of hidden laxative and diuretics abuse. Answer D is incorrect because neither the clinical exam nor the venous blood gas suggest hyperventilation. Answer E is incorrect because there is no suggestion of hyperadrenergic state on exam.
A 34-year-old woman presents with a complaint of fatigue, weakness, and cramping. She reports no significant medical history. She does not take prescription or over-the-counter medications or herbal supplements except for an oral contraceptive pill. She denies vomiting, diarrhea, and alcohol abuse.
On examination, her BP is 105/75 mm Hg, heart rate 73/min, temperature 36.7°C, and oxygen saturation 97% while breathing room air. Physical examination is remarkable for mild muscular weakness in the hips and shoulders. Rare fasciculations are observed.
Laboratory investigations include the following:

Hypomagnesemia persists despite ongoing replacement with oral magnesium oxide 800 mg three times daily. Further increases in magnesium dose have been limited by diarrhea.
Which of the following is the NEXT BEST STEP in management?
A. Hydrochlorothiazide
B. Cetuximab
C. Amiloride
D. Indomethacin
E. Furosemide
What is (C)
This patient with hypomagnesemia associated with Gitelman syndrome should be treated with amiloride.
The first step in the evaluation of a dysmagnesemia is to calculate the fractional excretion of magnesium (FEMg) to determine renal magnesium (Mg) handling:
FEMg = (urine Mg ÷ urine creatinine)/([serum Mg × 0.7] ÷ serum creatinine) × 100%
The serum Mg is multiplied by 0.7 to discount Mg bound to albumin. In the presence of hypomagnesemia, FEMg >2% indicates renal Mg wasting. In this case:
FEMg = (3.5 mEq/L ÷ 96 mg/dL)/([1.1 mEq/L × 0.7] ÷ 0.62 mg/dL) × 100% = 2.9%
Inappropriate renal magnesium excretion in the setting of hypomagnesemia, in addition to hypokalemia, metabolic alkalosis, and undetectably low urine calcium, are consistent with a diagnosis of Gitelman syndrome. Clinical manifestations commonly include fatigue, weakness, and cramping.
Amiloride is a selective blocker of the epithelial sodium channel (ENaC). ENaC blockers are thought to increase magnesium absorption based on their alteration of transepithelial voltage. When ENaC is blocked, cells in the distal nephron are unable to absorb sodium, creating a positive luminal and negative intracellular charge. This electrical gradient favors magnesium absorption through transient receptor potential melastatin-6 and -7 (TRPM-6 and TRPM-7) channels. Aldosterone antagonists are felt to work in a similar manner. Thus, in patients with persistent hypomagnesemia, a potassium-sparing diuretic may help increase serum magnesium levels.
A 61-year-old-man with heart failure with reduced ejection fraction (HFrEF) due to ischemic cardiomyopathy presents to the emergency department due to worsening dyspnea and lower extremity edema. He has a history of coronary artery disease status post left anterior descending artery stenting 6 months ago. Outpatient medications include clopidogrel, aspirin, atorvastatin, sacubitril/valsartan, and furosemide.
On examination, his BP is 101/60 mm Hg, heart rate is 62/min, respiratory rate is 20/min, oxygen saturation is 91% while breathing room air, and weight is 89 kg (baseline is 80 kg). He has jugular venous distension, bibasilar rales, and bilateral 3+ pitting edema to posterior thighs.
He is treated with furosemide IV twice daily and produced 2.2 L of urine in the first 24 hours. The next day, despite doubling the dose of furosemide IV, urine output decreased to 1.5 L/day, with persistent 3+ edema, bibasilar rales, and unchanged supplemental oxygen requirement. His weight has decreased to 88.3 kg at 48 hours.
Laboratory data are as follows:

UA is negative for blood and protein. Urine microcopy demonstrates scattered hyaline casts and no RBCs or WBCs.
Ultrasound demonstrates normal echogenic kidneys with no evidence of hydronephrosis.
Which ONE of the following is the MOST appropriate next step in management?
A. Prescribe empagliflozin
B. Change to furosemide infusion
C. Prescribe tolvaptan
D. Initiate dopamine infusion
E. Initiate ultrafiltration
What is (A)
Therapy with empagliflozin, a sodium-glucose co-transporter 2 inhibitor (SGLT2i), should be initiated for this patient with acute decompensated heart failure (ADHF) and type 1 cardiorenal syndrome.
Multiple clinical trials have shown that in patients with acute heart failure, the addition of SGLT2i to conventional diuretic therapy reduces all-cause mortality as well as subsequent heart failure events. Furthermore, SGLT2i therapy has been associated with a lower daily dose of loop diuretics and an improved volume of diuresis. In patients with HFrEF, SGLT2i treatment also is associated with reduced all-cause and cardiovascular death, and improved kidney outcomes. This patient remains volume overloaded with inadequate decongestion despite treatment with increasing dose of IV furosemide, and he would benefit from the additional of empagliflozin.
A 67-year-old man presents for 3-month follow-up on CKD secondary to hypertension. Home medications include losartan, spironolactone, furosemide, aspirin, and finasteride. On previsit laboratory tests, you note that his GFR fell from 44 to 30 ml/min. He denies any recent hospitalizations or acute illnesses. Review of systems is positive for intermittent headache and blurry vision. He denies chest pain but does note occasional palpitations. He denies shortness of breath but does endorse “smoker’s cough” in the mornings before his first cigarette. Vital signs are HR 80 and BP 120/78. Physical exam reveals a morbidly obese male in no acute distress. Heart rate is regular, and there are no rubs or murmurs. Breath sounds are clear. There is no edema on exam.
What is the most appropriate next step in the management of this patient?
A. Continue current plan of care; BP is at goal on current therapy
B. Reduce furosemide dose; his falling GFR and lack of edema suggest he is overdiuresed
C. Order ambulatory BP monitoring for this patient to evaluate control at home
D. Refer to cardiology for evaluation of his complaint of headache and palpitations
What is (C)
This patient is presenting with controlled office BPs, but no out-of-office readings are reported. However, he is showing evidence of hypertension-mediated organ damage, including worsening kidney function, neurologic symptoms of dizziness and blurred vision, and cardiac symptoms. Additionally, he is obese and a smoker. When taking the full clinical picture into consideration, there should be strong suspicion for masked uncontrolled hypertension, and home BP readings should be obtained.
A 24-year-old male comes in with resistant hypertension. He reports that he is on amlodipine 10 mg daily, losartan 100 mg daily, hydrochlorothiazide 25 mg daily, and spironolactone 50 mg daily. Patient reports following a low-sodium diet. He does not use any nonsteroidal anti-inflammatory medications, and he reports compliance with medications. Patient reports that he was diagnosed with high blood pressure about 8 years prior.
Vitals: Blood pressure 152/98, pulse: 64. Physical exam is unremarkable, and patient looks euvolemic.

What is the next best in management of this patient?
A. Recheck renin and aldosterone off spironolactone.
B. Check urine chloride.
C. Stop spironolactone and change to amiloride
D. Check serum ACTH and cortisol
What is (C)
The patient has hypokalemia and metabolic alkalosis in the setting of
euvolemia and hypertension at a young age, which is very suggestive of monogenic
hypotension.
Answer B is incorrect. A urine chloride would not help any further, because it would be
expectantly high in this case. Normally, plasma renin activity and aldosterone concentration
should not be checked on spironolactone, because it can falsely elevate both values, but, in this
case, both are suppressed, which suggests that values can be used; thus answer A is not the best
answer. This narrows the differential to Liddle syndrome, Cushing disease or syndrome, apparent
mineralocorticoid excess or licorice use, defect in the mineralocorticoid receptor, congenital
adrenal hyperplasia, or exogenous mineralocorticoid use. Answer D is not the best answer. The
patient does not exhibit any cushingoid signs, and serum glucose is normal. Similarly, there is no
evidence of congenital adrenal hyperplasia on physical exam. Mineralocorticoid receptor
mutation often presents in women and worsens in pregnancy. Licorice use and apparent
mineralocorticoid excess should be evaluated in this patient; however, seeing whether there is a
response to amiloride that would be consistent with Liddle syndrome in addition to screening
for licorice use would be a cost-effective measure prior to sending expensive genetic studies;
thus answer C is the best answer. The correct answer is discussed in the section on Management
in the setting of elevated urine chloride and hypervolemia.
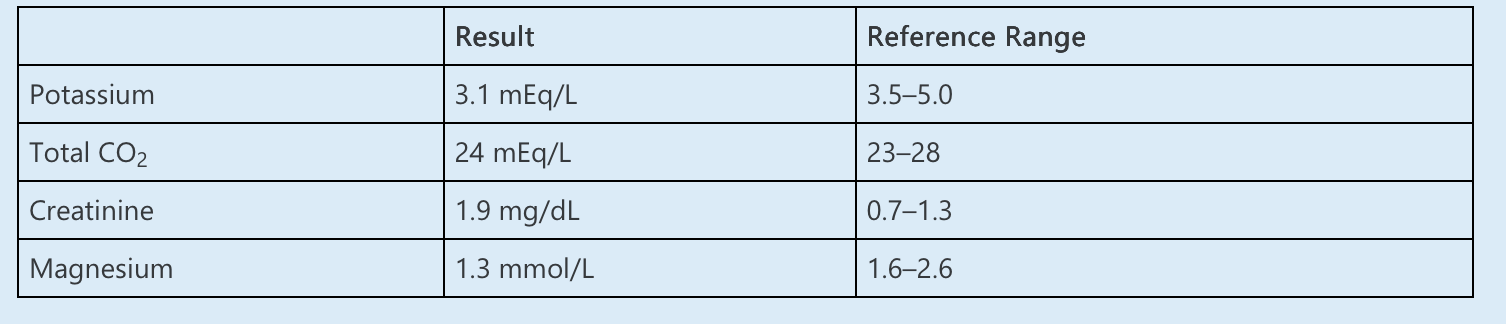
A 35-year-old man is referred to nephrology clinic for evaluation of hypokalemia and hypomagnesemia. He developed type 2 diabetes at age 11 years. His blood sugar is well controlled. He has no history of nephrolithiasis, gout, or liver disease. Family history is significant for ESRD in the patient’s father and paternal aunt.
His BP is 138/76 mm Hg, and the remainder of the physical examination is normal.
Laboratory studies are normal except for the following:
CT scan of the abdomen reveals numerous small cysts along the corticomedullary junction of both kidneys and an atrophic pancreas.
A defect in which of the following is MOST likely cause of this patient’s clinical syndrome?
A. Hepatocyte nuclear factor-1ß
B. Claudin-16/paracellin-1
C. H+-ATPase
D. Fibrocystin/polyductin
E. NaCl cotransporter
A defect in hepatocyte nuclear factor-1β (HNF1β) is the most likely cause of this patient’s presentation characterized by hypomagnesemia, CKD, and early onset of type 2 diabetes mellitus. HNF1β is a transcription factor that is involved in the development of the kidney, urogenital tract, pancreas, parathyroid glands, brain, and liver; therefore, mutations in the gene for this transcription factor have a variable phenotype and may include multiple abnormalities. The most common presentation includes early-onset type 2 diabetes along with pancreatic atrophy, hypokalemia, and hypomagnesemia. This disorder was previously called maturity-onset diabetes of youth (MODY) type 5 or “renal cyst and diabetes syndrome.” This condition may mimic Gitelman syndrome because of hypomagnesemia and hypokalemia.
Mutations in the gene encoding HNF1β is one of several disorders recognized recently that are characterized by autosomal dominant non-glomerular kidney disease, which results in progressive tubulointerstitial fibrosis and progression to ESRD. This family of disorders has been reclassified as autosomal dominant tubulointerstitial kidney disease (ADTKD). Typical features of these disorders include autosomal dominant inheritance, progressive CKD, bland urinary sediment, absent or mild proteinuria, and absence of hypertension in the early stages of disease. A renal concentrating defect is typically present. Histopathology usually features interstitial fibrosis, tubular atrophy, tubule microcysts, and basement membrane lamellation; immunofluorescence is negative for immunoglobulins and complement. ADTKD subtypes are further classified by the causative gene mutation.
This patient is a 48-year-old man with CKD stage 3A, type 2 diabetes, and resistant hypertension. Medications include losartan 80 mg, nifedipine 60 mg twice daily, and chlorthalidone 50 mg daily. You have tried spironolactone and eplerenone in the past, but he developed unacceptable side effects. You discuss the possibility of aprocitentan, and he wishes to know the side effects.
What is the most common reported side effect of aprocitentan?
A. Nausea and diarrhea, which tends to abate with time
B. Edema
C. Anaphylaxis
D. Hyperkalemia
What is (B)
In a multicenter blinded randomized phase 3 trial (PRECISION), the benefit of aprocitentan was compared with placebo among 730 individuals. Edema was the most commonly reported side effect, and occurred in 9% and 18% of individuals receiving aprocitentan 12.5 and 25 mg daily, respectively, versus 2% in individuals receiving placebo.
Seven patients were withdrawn from the study because of edema. Another reported side effect was lowering of hemoglobin. Gastrointestinal symptoms were not prominent in PRECISION, thus answer A is not the best answer. Although anaphylaxis is possible with any new agent, it is not commonly described with aprocitentan, and thus answer C is not correct. Answer D is incorrect.
Hyperkalemia is not a major feature associated with aprocitentan use, which suggests its potential utility over spironolactone and eplerenone for patients at risk for hyperkalemia.
This patient is a 72-year-old man with CKD stage 4, type 2 diabetes, and severe hypertension. When you see him in follow-up, he has a new diagnosis of heart failure with reduced ejection fraction. His losartan has been stopped, and he has been started on sacubitril-valsartan. Because you have stressed the importance of an ARB, such as losartan, on preservation of kidney function, he is concerned about the change in medication.
On the basis of the PARADIGM-HF trial, compared with an ACEI, what are the effects of an angiotensin receptor-neprilysin inhibitor?
A. More effective in reducing BP
B. More effective in reducing proteinuria
C. Faster decline in GFR
D. Greater risk of hyperkalemia
What is (A)
A post hoc analysis of the PARAGON-HF trial showed that sacubitril-valsartan compared with valsartan alone was more effective in reducing BP in heart failure with preserved ejection fraction patients with controlled hypertension, nonresistant hypertension, apparent resistant hypertension, and apparent MRA-resistant hypertension. Improved BP control with sacubitril-valsartan was subsequently shown in patients without heart failure who had essential hypertension.
None of the above trials reported significant adverse effects of AngII ARB and a neprilysin inhibitor (ARNI) compared to ARBs alone, with the exception of increased hypotension in the heart failure trials in patients with controlled hypertension at baseline.
A 28-year-old man is referred to an outpatient hypertension clinic for uncontrolled hypertension. He was diagnosed with hypertension in his early teens and has been prescribed lisinopril, amlodipine, and chlorthalidone for last 5 years. Despite excellent adherence to medications, his BP averages 160/90 mm Hg on a home log. He has been experiencing occipital headaches intermittently for the past 2 months but otherwise feels well. He does not report any illicit drug use. He drinks alcohol socially once every 2 weeks, and his last drink was 11 days ago. He does not use regular NSAIDs or other over-the-counter medications. His father died of a hemorrhagic stroke at the age of 43 years. His mother and siblings are healthy.
On physical examination, his BP is 182/98 mm Hg, heart rate is 120/min, and temperature is 37.2°C. His body mass index is 23 kg/m2. He is alert and oriented. Jugular venous distension is absent. He is tachycardic with no murmurs and an S4 sound on cardiac examination. Lungs are clear. The abdomen is soft with normal bowel sounds. Extremities show no edema. Neurologic examination is nonfocal.

EKG reveals sinus tachycardia and nonspecific T-wave changes. Chest x-ray is normal. Kidney ultrasound with duplex Doppler examination is negative for renal artery stenosis.
Which ONE of the following is the MOST LIKELY CAUSE of hypertension in this patient?
A. Aldosterone-producing adenoma
B. Liddle syndrome
C. Glucocorticoid-remediable aldosteronism
D. Syndrome of apparent mineralocorticoid excess
E. Cushing syndrome
What is (C)
The most likely cause of hypertension in this patient with hypokalemia, metabolic alkalosis, and elevated 24-hour urinary 18-oxocortisol and 18-hydroxycortisol is glucocorticoid-remediable aldosteronism (GRA).
GRA is the most common monogenic cause of hypertension and is an autosomal dominant, hereditary form of primary hyperaldosteronism. It is caused by a chimeric gene translocation in which the ACTH-responsive 5′-promotor of the 11β-hydroxylase gene is fused to the coding region of the aldosterone synthase gene. This results in ectopic production of aldosterone in the cortisol-producing region of the adrenal gland (zona fasciculata) under the influence of ACTH, with resultant hyperaldosteronism and feedback suppression of renin release.
Patients with GRA may have significant clinical (early-onset hypertension, hypokalemia, and metabolic alkalosis) and biochemical (high serum and urinary aldosterone and suppressed plasma renin activity) findings, similar to those with an adrenal aldosterone-producing adenoma. However, half of patients with GRA do not have hypokalemia at baseline, although this finding may develop after thiazide initiation. A strong family history of subarachnoid hemorrhage or hemorrhagic stroke (due to intracranial aneurysm rupture) is highly suggestive of GRA. Elevated 24-hour urinary levels of 18-oxocortisol and 18-hydroxycortisol suggest GRA. Genetic testing may also be done to definitively diagnose this condition. GRA-related hypertension can be treated with corticosteroids, which suppress ACTH and thereby block the ACTH-driven production of aldosterone, or with direct mineralocorticoid receptor blockers such as spironolactone or eplerenone.
A 33-year-old man is evaluated for CKD. He has been treated for hypertension for the past 3 years. Family history is significant for several relatives with ESRD, including his father at age 28 years, two sisters at ages 18 and 35 years, and two paternal aunts at ages 27 and 29 years. His only prescribed medication is lisinopril 5 mg daily, and he does not use NSAIDs or other nonprescription medications or herbal products.
Laboratory data are as follows:

The urine sediment is notable for a rare acanthocyte (0–2/HPF).
An MRI of the abdomen is shown below. The left kidney is 9.6 cm, and the right kidney is 10.5 cm, with no hydronephrosis. Multiple small, bilateral, medullary cysts are present (arrow), numbering at least 15 on the left and 5 on the right. Rare cortical cysts are also present. All cysts measure ≤1 cm in diameter.

Which of the following additional findings would be MOST CONSISTENT with this patient’s diagnosis?
A. Nephrocalcinosis
B. Interstitial fibrosis and tubular atrophy
C. Hepatic fibrosis
D. Hearing loss
E. Hepatic and pancreatic cysts
What is (B)
The most likely additional finding would be interstitial fibrosis and tubular atrophy on kidney biopsy in this patient with features of autosomal dominant tubulointerstitial kidney disease (ADTKD).
Defects in several genes have been linked to ADTKD, most commonly UMOD, REN, and MUC-1, less often HNF1B and SEC61A1. All types of ADTKD are associated with small corticomedullary kidney cysts, mild hypertension, and progressive CKD with a bland urine sediment and minimal proteinuria. ESRD occurs in an autosomal dominant, early-onset pattern. Associated characteristics are shown in the table below. ADTKD is usually diagnosed clinically, although genetic testing may be done in certain circumstances. Biopsy may be performed if family history is unavailable or inconclusive for ADTKD.
A 57-year-old woman was admitted to the hospital for partial colectomy due to refractory diverticulitis. She underwent surgery without complication. She is now receiving IV piperacillin/tazobactam 3.375 g three times daily. She has a history of type 2 diabetes and hypertension, for which she takes metoprolol and insulin. Review of systems is significant for decreased oral intake as well as diarrhea since surgery.
On physical examination, her BP is 127/63 mm Hg, pulse 78/min, and body mass index 24 kg/m2. The abdomen is mildly tender consistent with her recent surgery; sparse bowel sounds are present. She has no swelling in the extremities. The rest of the examination is normal.
Despite IV supplementation, her serum potassium concentration has progressively declined over the past few days. In the prior 24 hours, she received a total of 80 mEq of IV potassium chloride.
Laboratory data are as follows:

Urine output was 2.5 L in the prior 24 hours. Urinalysis shows specific gravity of 1.010, pH 7.0, no blood, and no protein. Urine potassium is 22 mEq/L and urine creatinine 40 mg/dL.
Which ONE of the following is the MOST likely cause of hypokalemia?
A. Decreased oral intake
B. Diarrhea
C. Metoprolol
D. Insulin
E. Piperacillin/tazobactam
What is (E)
This patient has urinary potassium loss due to piperacillin/tazobactam. It is thought that piperacillin causes hypokalemia and metabolic alkalosis by the same mechanism as that of penicillin. Two hypotheses have been put forward for explaining the mechanism of hypokalemia. First, piperacillin behaves as a non-reabsorbable anion, enhancing transepithelial electronegativity in the distal nephron and resulting in increased distal sodium delivery and potassium excretion. Second, the large amount of sodium administered with piperacillin increases distal delivery of sodium and excretion of potassium through the renal outer medullary potassium (ROMK) channel. Lastly, the solute diuresis causes a high flow rate in the cortical collecting duct and potassium excretion through maxi-K channels.
Evaluation of urinary potassium excretion is best accomplished by measuring potassium excretion in a 24-hour urine collection. Excretion of more than 25–30 mEq of potassium per day indicates inappropriate renal potassium loss. Here, the urine output of 2.5 L × 22 mEq/L is 55 mEq/d, which is inappropriately high. Measurement of the potassium and creatinine concentrations in a spot urinalysis is an alternative if collection of 24-hour urine is impractical. A spot urine potassium-to-creatinine ratio >15 mEq/g creatinine (or 1.5 mEq/mmol) usually indicates inappropriate renal potassium loss. Here, the urine K/Cr ratio is 22 mEq/L ÷ 0.4 g/L = 55 mEq/g, which is inappropriately high.
An 85-year-old man with a past medical history of CKD stage 3, hypertension, and coronary artery disease presents to the nephrology clinic for a follow-up visit. He reports recurrent episodes of dizziness and lightheadedness that began 6 months ago, when his antihypertensive regimen was intensified because of ambulatory BP measurements that averaged 160/90 mm Hg. These symptoms have prevented his participation in physical activity because of fear of falling.
His current medications include carvedilol, torsemide, and losartan. The dose of losartan was increased from 50 to 100 mg daily 6 months ago.
Today, his office BP is 148/88 mm Hg, and he reports dizziness upon walking to the examination table. He has trace bilateral lower extremity edema. The remainder of his examination is unremarkable.
Which one of the following is the NEXT BEST step in diagnosis?
A. Abdominal fat pad biopsy
B. Brachial artery palpation after BP cuff insufflation
C. 24-Hour ambulatory BP monitoring
D. Echocardiography
E. Tilt table testing
This patient, with symptoms of hypotension but paradoxical hypertension by standard BP measurement, should have Osler’s maneuver performed.
Osler’s maneuver involves inflation of the BP cuff to a level higher than systolic BP followed by palpation of the radial or brachial artery. If the artery is pulseless but palpable, the patient is classified as being “Osler positive.” Patients who are Osler positive are likely to have pseudohypertension, in which BP readings are falsely elevated due to sclerosis of the large arteries. One study reported differences of 10–54 mm Hg between BP measured by sphygmomanometry compared with BP measured by intra-aortic catheter in these patients. Falsely elevated BP readings are attributed to sclerotic, poorly compliant, uncompressible large arteries. Patients with pseudohypertension are frequently older and have coexisting coronary artery disease.
Abdominal fat pad biopsy may be useful in diagnosing systemic amyloidosis, which can cause orthostatic hypotension due to autonomic dysfunction. However, this is an invasive test and should only be undertaken in the presence of other signs and symptoms of amyloidosis. Tilt table testing and echocardiography, which may be used to diagnose autonomic dysfunction and cardiac abnormalities, respectively, would not be warranted until pseudohypertension is ruled out. Osler’s maneuver would be the next best step in diagnosis.
A 65-year-old woman with a history of alcohol use disorder presents with a 2-day history of abdominal discomfort and a 1-day history of nausea, vomiting, and dyspnea. She does not have a headache, changes in vision or hearing, chest pain, diarrhea, or urinary symptoms. She acknowledges that she often drinks alcohol to the point of intoxication, but she abstained from alcohol the previous day because of abdominal pain. She does not take any prescription or over-the-counter medications.
Her BP is 112/68 mm Hg and heart rate 92/min. Heart and lungs are normal. She has mild, diffuse abdominal tenderness to palpation but no rebound. There is no edema.
The following laboratory results are obtained:

Urinalysis demonstrates specific gravity of 1.004, pH of 5.0, and no blood or protein. Ketones are strongly positive. There are no crystals identified in the sediment.
Which of the following is the NEXT BEST STEP in management?
A. Hemodialysis
B. Fomepizole
C. Insulin and 0.9% saline
D. Thiamine and dextrose
E. 0.9% saline
What is (D)
The most appropriate initial treatment for this patient presenting with features of alcoholic ketoacidosis (AKA) is the administration of thiamine and dextrose.
This patient has an elevated anion gap metabolic acidosis (AGMA) in combination with an osmolal gap (OG) in the setting of recent abstinence from chronic alcohol consumption.
The OG may be calculated as follows:
- Measured osmolality – calculated osmolality = OG
- 315 mOsm/kg H2O – (Na × 2 + BUN/2.8 + glucose/18)
- 315 mOsm/kg H2O – (137 × 2 + 15/2.8 + 195/18)
- 315 – 290 mOsm/kg H2O= 25 mOsm/kg
- In this case, the OG is 25 mOsm/kg H2O.
By dividing by its molecular weight (58 mg/mOsm) and converting dL to kg, the contribution of acetone to the OG can be estimated:
(150 mg/dL × 10 dL/kg) ÷ 58 mg/mOsm = 25.9 mOsm/kg H2O
The index of suspicion must remain high for toxic alcohol poisoning in a patient presenting with AGMA and an elevated OG, but this patient does not demonstrate the visual disturbances that occur with methanol poisoning or the AKI and calcium oxalate crystalluria seen with ethylene glycol toxicity. In this case, acetone accounts for the OG, and it is not necessary to invoke the presence of a toxic alcohol that would indicate the need for fomepizole or HD.
Profound ketoacidosis can develop in patients with chronic alcohol use. Contributing factors include poor nutritional status, which results in a reduction in the insulin-to-glucagon ratio, leading to accelerated fatty acid utilization as a source of energy. Lower insulin levels decrease carnitine acyltransferase activity, impairing the entry of fatty acids into mitochondria, where they would normally enter the citric acid cycle, and instead shunting them to ketone production. Chronic metabolism of ethanol in the liver leads to an increased NADH/NAD+ ratio, causing a mass balance shift of acetoacetate to β-hydroxybutyrate to a greater extent than typically seen in diabetic ketoacidosis (DKA). Patients most often present after a period of abstinence when the ethanol level is low, because withdrawal increases circulating catecholamines, provoking more peripheral mobilization of fatty acids.
Treatment of AKA includes infusion of dextrose, which stimulates insulin release, which in turn lowers peripheral fatty acid mobilization and hepatic output of ketoacids. When giving dextrose to patients who have alcohol use disorder, or any condition prone to malnutrition, it is important to also ensure that thiamine is given beforehand. Thiamine is an essential cofactor in the citric acid cycle and therefore necessary for energy production. In the absence of adequate thiamine, pyruvate accumulates and is reduced to lactic acid. Furthermore, the administration of dextrose can provoke consumption of depleted thiamine stores, potentially resulting in Wernicke encephalopathy. Note that patients with AKA may present with a normal, low, or, as in this case, modestly elevated blood glucose.
A 30-year-old man has ADPKD determined to be
Mayo image class 1D based on the HtTKV from an
MRI imaging. His serum creatinine is 1.0 mg/dL. The
nephrologist discusses with the patient that he will be
benefit from treatment with tolvaptan.
In the countries where a vasopressin V2R antagonist
has been approved for the treatment of ADPKD,
which of the following patients is likely to derive
the most benefit from treatment with this drug?
A. 55-year-old man with HtTKV 300 mL/m.
B. 35-year-old woman with PKD2 mutation and
serum creatinine 0.9 mg/dL.
C. 30-year-old man with HtTKV 1,200 mL/m and
serum creatinine 0.9 mg/dL.
D. 70-year-old man with serum creatinine 2.6 mg/dL.
E. 40-year-old woman with TKV 1,800 mL and three
cysts accounting for most of the volume (520, 464,
and 285 mL)
What is (C)
Patients who received tolvaptan in their early
disease state likely gain more benefit compared with patients with declined kidney function.
According to baseline GFR at time of treatment initiation, tolvaptan might delay reaching stage
5 CKD by 7.3, 4.4, 2.9, or 1.5 years if baseline eGFR was 90, 60, 45, or 30 mL/min, respectively.
These extrapolations are made using the average decline in eGFR between placebo (−3.7 mL/min per year) and tolvaptan (−2.72 mL/min per year) groups in the TEMPO3:4 trial. Patients from answer A and B do not meet the criteria for high-risk patients, and the patient from answer E is likely atypical ADPKD. Because of his age and advanced CKD, the patient from answer D is not likely to benefit from tolvaptan
A 36-year-old woman with Crohn disease is admitted to the hospital for a flare and undergoes small bowel resection with ostomy placement. During the first 24 hours after surgery, ostomy output is excessive. Subcutaneous octreotide is initiated with moderate reduction in stool volume by the third day. She reports incisional pain.
Medications include morphine as needed, methylprednisolone, lisinopril, and heparin subcutaneously.
Her BP is 168/92 mm Hg and heart rate 104/min. She has normal heart and lung examinations. Her abdomen is tender consistent with her recent surgery. The surgical wound and ostomy appear healthy.
The following laboratory data are obtained:
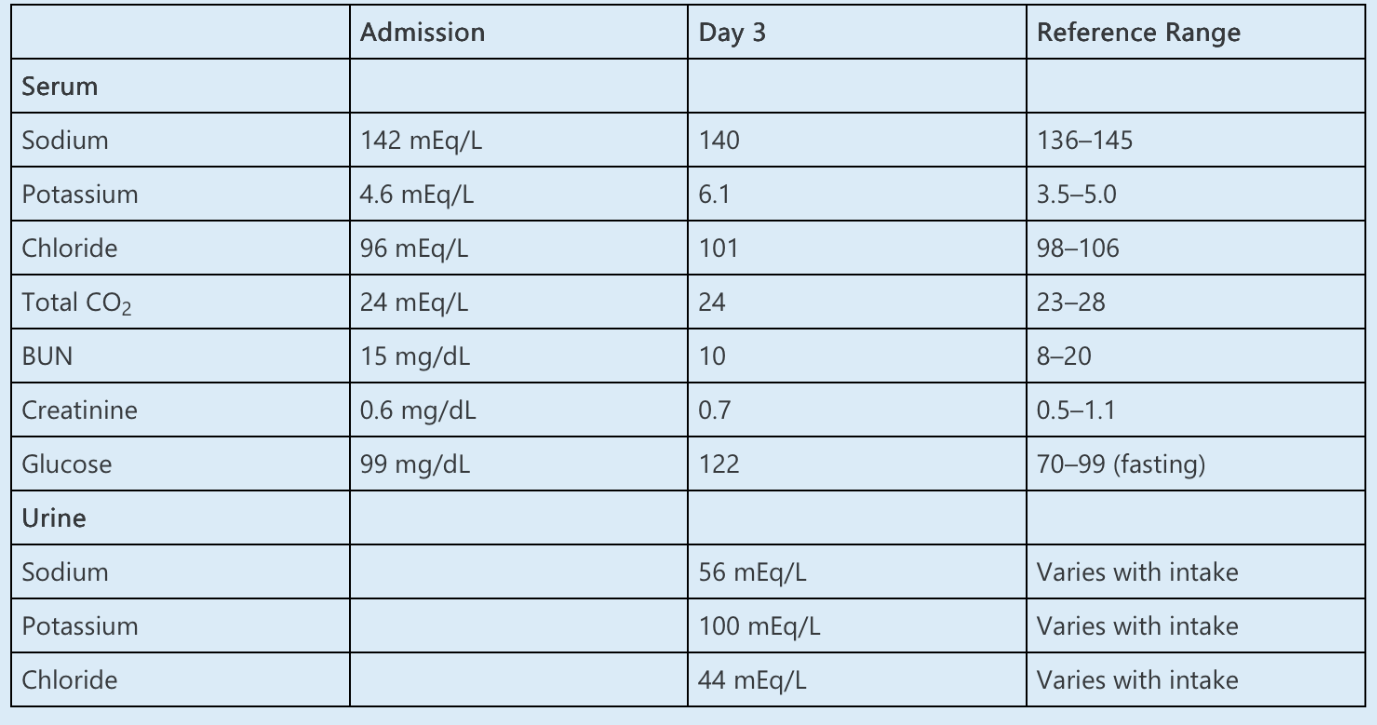
The patient is treated with 10 units of regular insulin and glucose, and repeat serum potassium is 4.9 mEq/L.
Which of the following is the MOST LIKELY cause of the hyperkalemia?
A. Metoprolol
B. Lisinopril
C. Heparin
D. Octreotide
E. Methylprednisolone
What is (D)
This patient is hyperkalemic because of relative insulin deficiency induced by octreotide. Insulin deficiency results in hyperkalemia by reducing the activity of Na-K-ATPase, resulting in reduced cellular uptake. Octreotide is a potent inhibitor of insulin secretion, and patients can experience up to a 1 mEq/L increase in serum potassium after its administration. This effect can be more pronounced in patients with ESRD. Patients with relative insulin deficiency, as can occur in inflammatory bowel disease and undernourishment, are also at greater risk. If discontinuation of octreotide is not feasible, patients may require exogenous insulin. Hyperkalemia in this case responded readily to exogenous insulin challenge.
Lisinopril and heparin both cause hyperkalemia by decreasing the production of aldosterone. Heparin inhibits aldosterone synthase in the zona glomerulosa. Lisinopril inhibits ACE, decreasing the angiotensin II-mediated stimulus for aldosterone production. Hypoaldosteronism results in a reduction in renal potassium excretion. The urinary potassium concentration is of limited utility in the evaluation of hyperkalemia, but low concentration (urinary potassium concentration of <40 mEq/L or a urine potassium-to-creatinine ratio of <20 mEq/g) is consistent with impaired renal potassium excretion. In this case, the urine potassium concentration of 100 mEq/L suggests that renal potassium excretion is intact, which indicates that neither the lisinopril nor the heparin is the primary cause of hyperkalemia.