The 3 subatomic particles of an Atom
What is Protons, Neutrons, and Electrons
Subatomic Particle responsible for Isotopes
What is neutrons
Elements in the same group share this.
What are the same number of valence electrons
The three classes of elements are these.
What are metals, metalloids, and nonmetals?
The modern periodic table is organized according to this.
What are by increasing atomic number?
The Atomic Number is the number of .....
What is protons
Ions are created when....
What is gaining or losing electrons?
3 Periodic Trends
Atomic Radius, Ionization Energy, Electron Affinity/Electronegativity
These are the properties of metals.
What are conductivity, high melting points, most are solids at room temperature, shiny, ductile, and malleable?
Tell us the number of energy levels/shells
What are periods?
Atomic Mass is = 12, Atomic Number is 6, Number of Neutrons =?
What is 6
The top number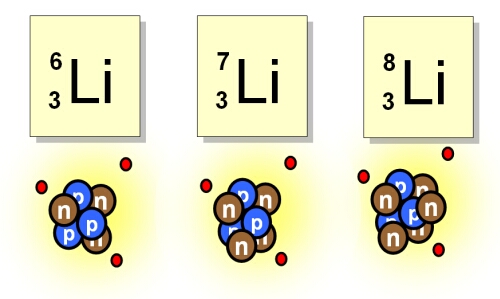
What is the atomic mass
The energy required to remove a valence electron from an atom in its gaseous state.
What is Ionization energy?
These are the properties of nonmetals.
What are brittleness, low boiling/melting points, dull, and are bad insulators?
The columns on a periodic table are called this.
What are groups?
The first energy level/shell/orbital location is
What is closest to the nucleus?
An atom with a completely full outer most (valence) energy level is this.
What is unreactive/stable?
Why does the atomic radius increase as you go down a group?
More orbitals; less attraction between the valence electrons and protons in nucleus.
These gain an electron
What is Non-metals
Extremely reactive elements in group 1 have only 1 valence electron and are known as this.
What are alkali metals?
A majority of an Atom's mass is located
What is the Nucleus
A Sodium Ion with 11 Protons, 10 Electrons has what charge?
What is +1?
The most reactive metal on the periodic table
What is Francium
These are the properties of metalloids.
What are many properties of both metals and nonmetals?
Group 17 contains highly reactive nonmetals with seven valence electrons that tend to react violently with alkali metals and are more commonly known as this.
What are halogens?