This type of reaction has heat written as a product.
For example: A -> B + heat
Exothermic Reaction
What is needed for an effective collision?
Proper KE and Orrientaion
True or False: When pressure is increased, the reaction shifts towards the side with the fewest moles of gas.
TRUE: When pressure is increased, the reaction shifts towards the side with the fewest moles of gas.
True or false: △H is always positive if energy is a product.
FALSE: △H is always positive if energy is a reactant.
A + energy -> B △H = +
A -> B + energy △H = -
What is the difference between △H forward and △H reverse?
The sign.
What factor affects the rate of reaction for gases?
pressure
What does le chatelier's principle help us predict?
How a system will return to equilibrium if there is a disturbance to equilibrium.
C(s) + O2(g) --> CO2(g) △H –393.5
Cite evidence to justify a claim that the reaction is endothermic or exothermic.
The △H is a negative value. This means that energy is release, which means the reaction is exothermic.
Is this reaction favored in tems of enthalpy? Why?
Yes, the reaction is exothermic. Nature favors reaction that lose enthalpy (- delta H)
What can be said about the concentrations and rates of the forward and reverse reactions at equilibrium?
Concentration R&P: Constant
Rate F&R: Equal
A + B ⇌ C + D + Heat
What direction does the equilibrium shift if the temperature is increased?
A + B ⇌ C + D + Heat
Reverse/Left. Treat heat as a product, which is now in excess and needs to be used up to restore equilibrium.
Is there an entropy increase or decrease, and why?
2C(s) + H2(g) --> C2H2(g) △H = +227.4
entropy decreases. One mole of gas is more oredered than one mole of gas and two moles of solid.
Chaos! First person to correctly label all of the arrows for the forward reaction and bring their board to Mr. Ruffler to check wins the points for their group.
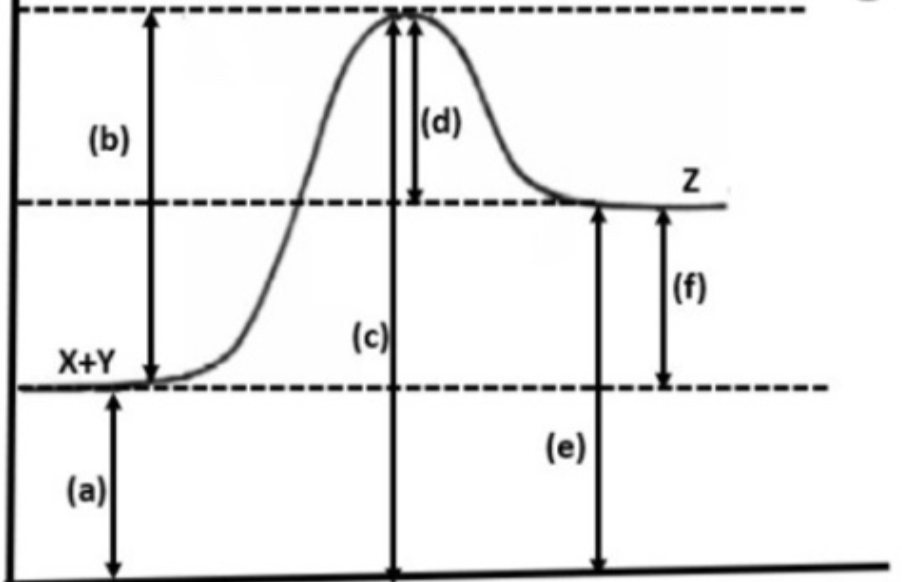
a) PE Reactants
b) Ea Reactants
c) Energy Activated Complex
d) N/A (Ea reverse)
e) PE Products
f) delta H
What does the universe favor in terms of enthalpy and entropy?
Low Enthalpy and High Entropy
"Lazy and Messy"
Which concentration (s) could be decreased to favor a shift forward?

decrease [H2O] only
How much energy is produced when 3.25 moles of C2H2 (g) are formed from it's elements?
2C(s) + H2(g) --> C2H2(g) △H = +227.4
△H = 3.25(+227.4) = +739.05 kJ
PE Reactants Forward: 40 kJ
△H Forward: - 20 kJ
PE Activated Complex: 80 kJ
What is the Activation Energy REVERSE?
60 kJ
Explain how a catalyst works and what it does.
A catalyst puts reactants in the proper orientation, thus LOWERING the activation energy.
Fe₃O₄(s) + H₂(g) ⇌ 3 FeO(s) + H₂O(g)
State what happens to the equilibrium if the pressure is changed from 760 mmHg to 600 mmHg
Fe₃O₄(s) + H₂(g) ⇌ 3 FeO(s) + H₂O(g)
No change. Equal moles (1 mol) of gas on each side.
What is △H when 20 mol of oxygen gas is formed from water at STP?
2H2(g) + O2(g) -> 2H2O(g) △H = –483.6
2H2(g) + O2(g) -> 2H2O(l) △H = –571.6
Water implies liquid, also at STP H2O is a liquid so use 2H2(g) + O2(g) -> 2H2O(l) △H = –571.6.
Flip the eqn and △H : 2H2O(l) -> 2H2(g) + O2(g) △H = +571.6
multiply △H by 20, and △H = + 11,432 kJ