When writing a procedure you must start each step in your procedure with a what?
Verb
What two subatomic particles are located inside the nucleus of an atom AND what are their charges?
Protons (positive) and Neutrons (neutral/no charge)
Define Isotope.
An atom that has the same number of protons but a different number of neutrons, therefore, the mass of the atom is different than the original atom/element.
In the following equation, what type(s) of radiation is occurring?
19277 Ir → ? + 42 He + 00 Ɣ
Alpha and Gamma
How do you define half-life?
The amount of time it takes for HALF of an element/isotope to decay into a stable element.
In this graph, which axis is the independent variable?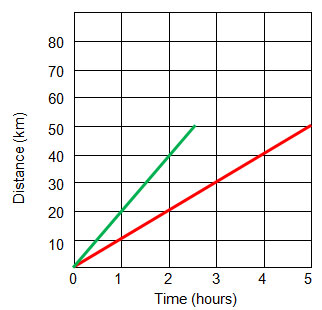
X-axis, time (hours)
What subatomic particle orbits the nucleus of an atom AND what is its charge?
Electrons (negative)
Which of the following IS an isotope?
Carbon (C) - 12
Iodine (I) - 126
Neon (Ne) - 20
Lithium (Li) - 7
Iodine (I) - 126
In the equation below, what did the element decay to? (Be sure to include mass and atomic number with the element.)
24797 Bk --> ? + 0-1 e
24798 Cf (Californium)
What is the half-life of an element that decays from 50 grams to 6.25 grams in 6 hours?
Half-Life (t1/2 = 2 hours)
What are the 5 things a graph must have?
2. Labeled x and y-axis
3. Scale
4. Key/Legend
5. Units
How many valence electrons does the atom Fluorine (F) have?
7
In the isotope Lead (Pb) - 210 calculate the following:
# of protons:
# of neutrons:
Atomic Mass:
82 protons
128 neutrons
Atomic Mass = 210
In the equation below, what was the element before it decayed?
? --> 19779 Au + 42 He
Element/Isotope: 20181 Ti
The half-life of a radioactive element is 2 seconds. You have 240g right now. How much will you after 12 seconds?
3.75 grams
Bob wants to try a new spaghetti sauce (Ragu) instead of using his normal spaghetti sauce (Prego) Bob eats Prego for one week and ranks the taste of his meals on a scale of 1-10. The next week Bob does the same thing with the Ragu sauce.
What is the.....
Control Group, Experimental Group, Independent Variable, and Dependent Variable.
Control: Prego
Experimental: Ragu
Independent Variable: Type of pasta sauce
Dependent Variable: Taste Ranking
Draw a shell model for the atom Nitrogen (N).

Find the average atomic mass for these isotopes. (Be sure to include your unit!)
Mercury (Hg) - 201 @ 70%
Mercury (Hg) - 205 @ 10%
Mercury (Hg) - 203 @ 20%
Average Atomic Mass = 201.8 amu
What type(s) of radiation occurred in the equation below?
16566 Dy --> 16164 Gd + ?
Alpha Radiation
Right now you have 2g of an unknown radioactive isotope. Your professor at Harvard told you its half-life is 6 years. How much did you have 36 years ago?
128 grams
Graph the following. (Remeber to include what a graph needs!) (Be careful to put the right numbers on the right axis!)
Heart Rate (BPM) Squat Sets (12 per set)
60 1
68 2
72 3
The teacher will draw the correct answer!
Answer the following about the atom Phosphorus (P).
How many protons, electrons, neutrons, electron shells, valence electrons, AND what is its atomic mass rounded to a whole number?
15 protons, 15 electrons, 16 neutrons, 3 electron shells, 5 valence electrons, and atomic mass is 31.
What is the average atomic mass (include unit) AND identify the unknown element/isotope?
X - 238 @ 99 %
X - 235 @ 1 %
X - 234 @ 0.006 %
Average Atomic Mass: 237.98404 amu
Element/Isotope: Uranium (U)
261102 No --> ? + 42 He + 00 Ɣ
What type(s) of radiation occurred?
What did the element decay to (include numbers)?
Is the new element stable or unstable?
How many neutrons does the new element have?
Type(s) of Radiation: Alpha and Gamma
Element: 257100 Fm
Stable or Unstable: Stable
Neutrons: 157N
Use the graph to determine the half-life of Radon (Rn) - 222.
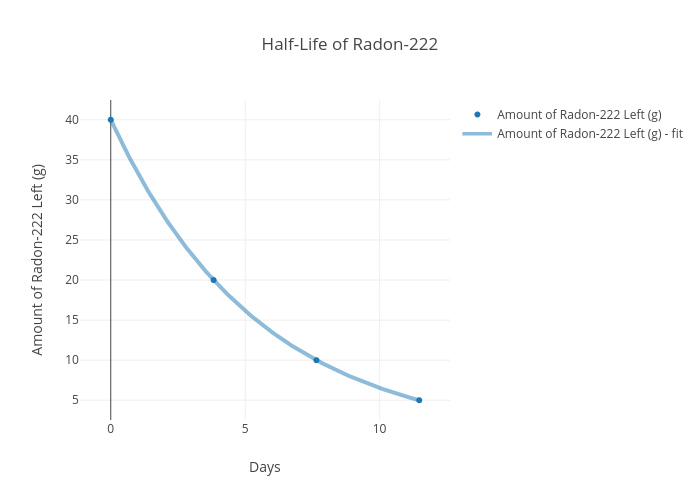
Half-Life (t1/2) = About 4 days