He
Helium
Which particle has a positive charge?
Proton
How many periods are in the periodic table?
7
How many atoms are in O2 ?
2
What is the atomic number of this atom?
2
P
Phosphorus
Which particle that has no charge?
Neutron
The atomic number is the number of ___ in the atom's nucleus.
Protons
How many atoms of each element are in NaCl?
Na=1, Cl=1
7
Fe
Iron
Which particles make up the nucleus of an atom?
Protons and Nucleus
Elements in the periodic table are listed in order of their ___.
Atomic Number
HCl has how many atoms of each element?
H=1, Cl=1

Nitrogen
Au
Gold
What does the atomic number represent?
The number of protons or electrons
The number found below the chemical name is the ___.
Atomic Mass
How many elements are in this compound?... NaHCO3
4
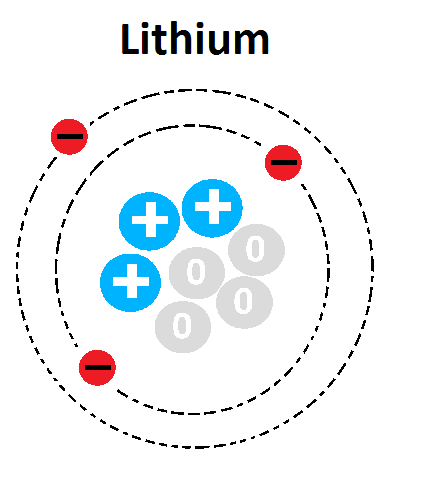
What element is represented in this Bohr Model?
Lithium
K
Potassium
What is the atomic mass for helium?
4
How many groups make up the periodic table?
18
4 NaCl represents several molecules of table salt. How many atoms of chlorine are present in this example?
4
An atom has an atomic number of 15 and a mass number of 31. How many protons are there in the atom?
15

