H
hydrogen
What is the charge of a proton?
positive
What is a row of the periodic table called?
a period
Would you find metals or non-metals in the top right area of the periodic table?
Non-metals
Is the appearance of metals shiny or dull?
Shiny
What does the atomic number represent?
Number of protons
These molecules are made up of the same type of atoms.
Elements
Which of the following images show an element?
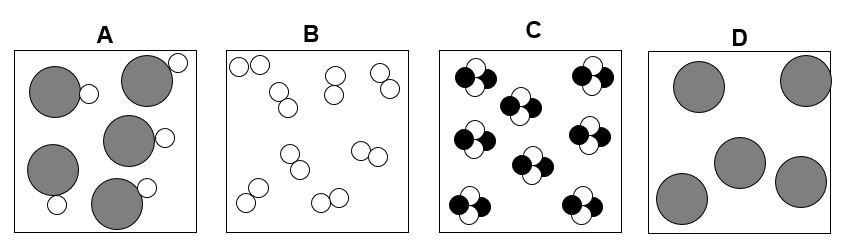
Images B and D
A change of state from a solid to a liquid is an example of a ____________ change.
Physical
Mg
magnesium
What is the charge of an neutron?
it has no charge
What is a column of the periodic table called?
a group
Which type of elements are found in a diagonal line between metals and non-metals?
Metalloids
Metals are good conductors of heat and electricity - True or False?
True
What does the mass number represent?
Number of protons plus neutrons in the nucleus
These molecules are made up different types of atoms.
Compounds
Which of the following images show a compound?
Images A and C
When solutions A and B are mixed, a gas is produced. This is evidence of a ____________ change.
Chemical
Cl
chlorine
What is the negative particle that orbits the nucleus of an atom?
electron
How many periods are there on the periodic table?
7
Which of the following elements is a metal?
Bromine, carbon, lithium, arsenic
Lithium
Which type of element has higher density - metals or non-metals?
Metals
What four things are included in an element's square?
atomic number, atomic mass, symbol and element name
Is H2O2 an atom, element, compound or mixture?
Compound
Which of the following images is a mixture of elements?

Image A
When a new substance is formed, this is the result of a _______________ change.
Chemical
Na
sodium
What particles are in the nucleus of an atom?
protons and neutrons
How many groups are on the periodic table?
18
Which of the following elements is a non-metal?
Fe, F, Mg, Na
F - fluorine
Which type of element exists as solid, liquid and gas at room temperature - metals or non-metals?
Non-metals
What is the atomic number, atomic mass and symbol for gold?
Atomic # is 79 Atomic mass is 196.967 or 197 Symbol is Au
Is C6H12O6 an atom, element, compound or mixture?
Compound (glucose)
Which of the following images is a mixture of compounds?

Image B
Temperature change is evidence that a ___________ change has occurred.
Chemical
K
potassium
Correctly identify the particles a, b and c.
a = electron
b = neutron
c = proton
How is the periodic table arranged?
by atomic number
Is the first element on the periodic table a metal, non-metal or metalloid?
Non-metal - hydrogen is the only non-metal on that side
One property of metals is they can bend without breaking. This means they are m______________.
malleable
The mass number is made up of
neutrons + protons.
Any two or more atoms bonded together are known as a...
molecule
Which of the following images is a mixture of elements AND compounds?

Image C
Which of the following is an example of a physical change?
A. cooking an egg
B. ice-cream melting
C. a candle burning
B. ice-cream melting