What which elements have the least amount of shells?
Hydrogen and Helium.
What is atomic radius?
The size of an atom.
This element has 1 valence electron and 1 shell.
Hydrogen.
What are valence electrons?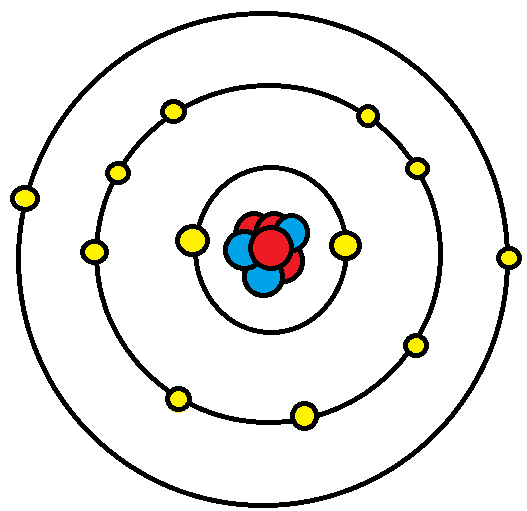
A valence electrons are electrons in the outer most energy level/shell.
What happens to the shells when you move down rows in the periodic table?
They increase by one.
What are the trends for Atomic radius?
Increases going down and left.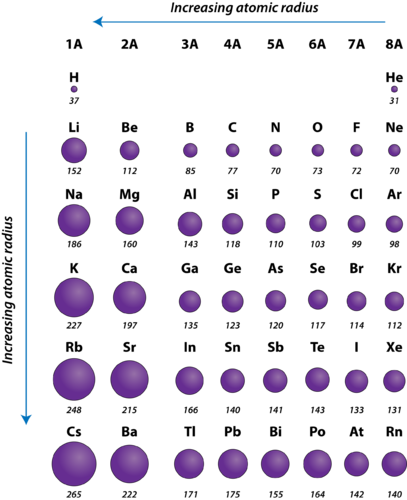
This element has 5 valence electrons and 4 shells.
Vanadium.
What direction do you move on the periodic table in order to increase the number of valence electrons in the outermost shell?
You move to the right.
How many shells does Sodium have?
3
What element has the least atomic radius?
Helium.
This element has 2 valence electrons and is the most electronegative in its column.
Beryllium. 
How many valence electrons does Titanium have in its outermost shell?
How many shells does Iron have?
4
What element has the most atomic radius?
Francium.
This element in the alkaine earth metal, forms a dark oxide-nitride layer.
Calcium.
Name a group that has 2 valence electrons on the outermost layer,
Alkaline earth metals, transition metals
What is the limit of shells on an element?
6
List these elements from the most to the least atomic radius.
Oxygen, Neon, Magnesium, Arsenic.
Magnesium, Arsenic, Oxygen, Neon.
This element is gas at a state of 0 c, has the most electronegativity in its column, 2 valence electrons, and 2 energy levels.
Oxygen. ![]()
True or false: Zinc has a high valence electron than Cobalt.
False.