What is the density (in g/mL) of a liquid that weighs 344 g and has a volume of 22 mL? Round to the nearest hundredths place.
15.64
A patient is to receive 63 mmol of potassium phosphate dibasic (MW 174). How many grams of potassium phosphate dibasic are represented by this dose? Please round your answer to the nearest hundredth.
Answer: ___ grams
10.96
How many grams of dextrose are used to prepare a 228 mL bag of D5W for intravenous infusion? Round to the nearest whole gram.
11
A pharmacist has a 45 g tube of 2% w/w ointment. The pharmacist needs a 0.5% ointment. How many grams of a nonmedicated ointment base should be added to the 2% ointment to achieve the desired concentration? Round your answer to the nearest whole number.
Answer: _____ grams
135
How many mL of Lantus (strength 100 unit/mL) would you need to give someone if their dose was 23 units? Do not round.
0.23
If 500 mL of a liquid weighs 3.8 kg, what is its density in g/mL? Round to the nearest tenths place.
7.6
How many millimoles of calcium chloride (MW 111) are present in 42 g of substance? Please round to the nearest tenth.
Answer: ___ mmol
A medication order for a patient calls for 32 mEq of potassium chloride (MW 74.5, valence 1) in 1000 mL D5W. How many milliliters of a 10% w/v concentrated potassium chloride stock solution should be added to the D5W? Round your answer to the nearest tenth.
23.8
A pharmacy receives a medication order for 0.7 L of 2.9% sodium chloride solution. The pharmacy only has sodium chloride available as a 0.9% solution and a 23.4% solution. How many milliliters of the 23.4% solution would be needed to make the compounded sodium chloride solution? Round your answer to the nearest whole number.
Answer: _____ mL
62
Eddy is to receive 260 mg of penicillin twice daily for 10 days for treatment of recurrent cholera. The penicillin in your pharmacy says that 250 mg = 400,000 units. How many units of penicillin will Eddy receive with each dose? Do not round
416,000
You need 62 grams of polysorbate 80 (s.g. = 1.08) to create a compound. Your supplier charges $114 for 500 mL of polysorbate 80. How much does the polysorbate required for the compound cost?
13.09
A 47 year old male is to receive 40 mEq of intravenous potassium chloride over 4 hours. How many mg/hr of potassium chloride (MW 74.5; valence 1) will the patient receive? Please round your answer to the nearest whole number.
Answer: ___mg/hr of KCl
745
A medication order calls for 3,572 mL of D5WNS to be administered over 6 hours. Calculate the quantity, in grams, of the sodium chloride administered in a 4 hour period. Round answer the the nearest tenth.
21.4
81 mL of a solution containing 70% v/v alcohol
72 mL of a solution containing 35% v/v alcohol
73 mL of a solution containing 15% v/v alcohol
And a sufficient quantity of a nonalcoholic solution to total 500 mL
Round your answer to the nearest whole number.
Answer: _____ % v/v
19
What is the % ionization of Valproic acid (pKa of 4.8), when it is in the stomach (pH=3)? Round to the nearest whole percent.
2
If 2.63 fl. oz. of glycerol weighs 74.1 g, what is its specific gravity? Round to the nearest hundredths place.
0.95
A 32 year old female just delivered a baby and is prescribed 1.4 grams/hr of magnesium sulfate for eclampsia. How many mEq of magnesium sulfate (MW 120; valence 2; # of particles 2) will the patient receive in 24 hours? Please round your answer to the nearest whole number.
Answer: ___ mEq
560
An IV bag containing 50mg/250mL of bumetanide is to be administered at a rate of 36mcg/min. Calculate the drip rate in drops/min if using a drip set of 30 drops/mL. Round your answer to the nearest whole drop.
5
How many milliliters of a 70% w/v solution of a medication should be used in preparing 58 mL of a stock solution such that 5 mL diluted to 50 mL will yield a 2% w/v solution of the medication? Round your answer to the nearest whole number.
Answer: _____ mL of 70% stock solution
17
What is the % ionization of estrone (pKa of 10.8) when it is mixed with milk of magnesia (pH=10)? Round to the nearest whole percent.
86
If 50 glycerin suppositories are made from the following formula, how many milliliters of glycerin, having a specific gravity of 1.25, would be used in the preparation of 30 suppositories?
Glycerin 91g
Sodium Stearate 9g
Purified Water5 g
44
Calculate the osmolarity (mOsm/L) of D5/ 1/2 NS (dextrose 5%/ 0.45% NaCl). Please round your answer to the nearest tenth.
NaCl: MW 58.5; valence 1; # of particles on dissociation 2
Dextrose: MW 180; valence 1; # of particles on dissociation 1
Answer: ____ mOsm/L
431.6
An order for a critical care patient with a 4L daily fluid limit is as follows:
- D5W to be given over 24 hours
- 66 mL IVPB antibiotic to be run over 30 minutes every 6 hours
Calculate the rate of D5W in mL/hr over the 24 hour period. Round the nearest whole number.
170
How many milliliters of a 100 mg/mL solution must you add to 27 mL of a 20 mg/mL solution to result in a 3% solution? Round to the nearest whole number.
Answer: _____ mL
4
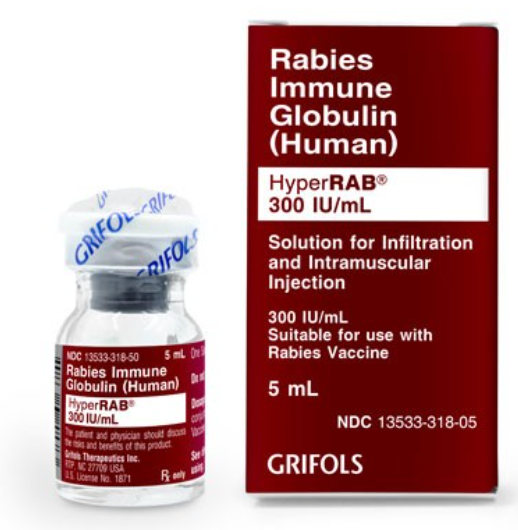
A 9.7 kg child is brought to your ER following a racoon bite. The physician is concerned about rabies and wants the child to receive HyperRab. You have the below vial in your pharmacy. The dose is 20 units/kg. How many mL of HyperRab should your patient receive? Round your answer to the nearest hundredths place.
0.65