What piece of lab equipment would we use to accurately measure liquid?
Graduated Cyclinder
Describe density
The amount of mass in a given object.
Describe a physical change
Alter the form, state, or appearance without changing it's chemical composition.
What is the periodic table?
Chart of elements going in increasing atomic number
Define force
A push or pull on an object

Tell me the measurements in each cylinder.
500, 25, 120, 225
Write down the formula for density
D=M/V
Explain how a chemical change is different from a physical change.
Chemical changes form completely new substances and usually there is evidence a chemical change has occurred.
Difference between an element and a compound
Element is pure and a single substance but a compound is made up of two or more elements.
Difference between balanced and unbalanced forces
Balanced force there will be no movement but in an unbalanced force it will move in the direction that has the stronger force
Name this piece of equipment and tell me how much the object weighs.

93.7
What units of measurement do we use for density?
g/mL
g/L
g/cm3
List at least 3 pieces of evidence a chemical change has occurred.
Gas, precipitate formation, color change, odor change, energy change (loss/gain heat)
Tell me 2 things you can determine about this element based on its card.

Answers vary (Nitrogen, atomic number 7, atomic mass 14, etc.)
Describe inertia and which of Newton's laws discusses inertia.
Tendency of an object at rest to stay at rest and vice versa.
Newton's first law
Explain the difference between an inference, observation, and measurement.
Inference - statement based on what you think or expect
Observation - statement made using your senses and what you observe
Measurement - exact measurement using various pieces of lab equipment.
What is the density of a material if a sample with a mass of 45.82 g takes up a volume of 8.75 mL? (Round to nearest tenth)
5.2 g/mL
Give 2 examples of a physical change.
:max_bytes(150000):strip_icc()/TC_608336-examples-of-physical-changes-5aa986371f4e1300371ebebb.png)
How can I tell the difference between the number of atoms and elements? Use the example below:
2H2O
Number of elements states how many elements there are total while number of atoms is referring specifically to how many of each element there are total.
Explain what Newton's 2nd law of motion states and give an example.
Acceleration, mass, and force
Fishing rod, car pulling a camper, etc.
Tell me the measurement of each item starting with the one at the top.

12, 19, 10, 5, 3.5, 8, 9, 14 all in cm
What mass would a sample of 32.6 cm3 of copper be? The density of copper is 8.96 g/cm3. (Round to nearest tenth)
292.1 g
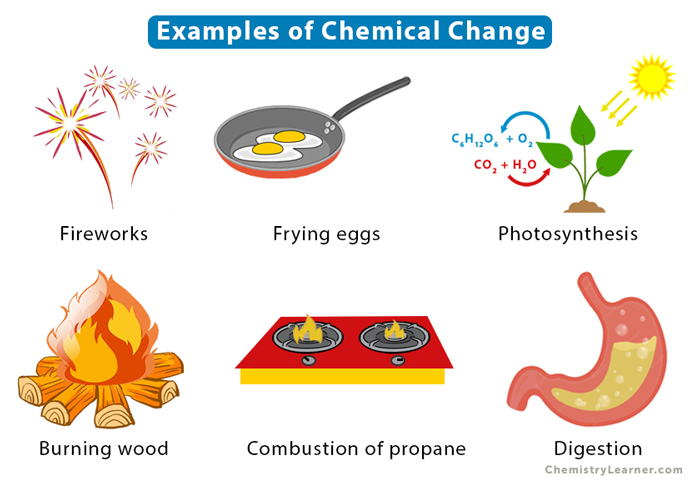
Give 2 examples of a chemical change.
The chart below lists samples of three materials. Classify each sample in the chart as an element, a compound, or a mixture by circling the correct classification.
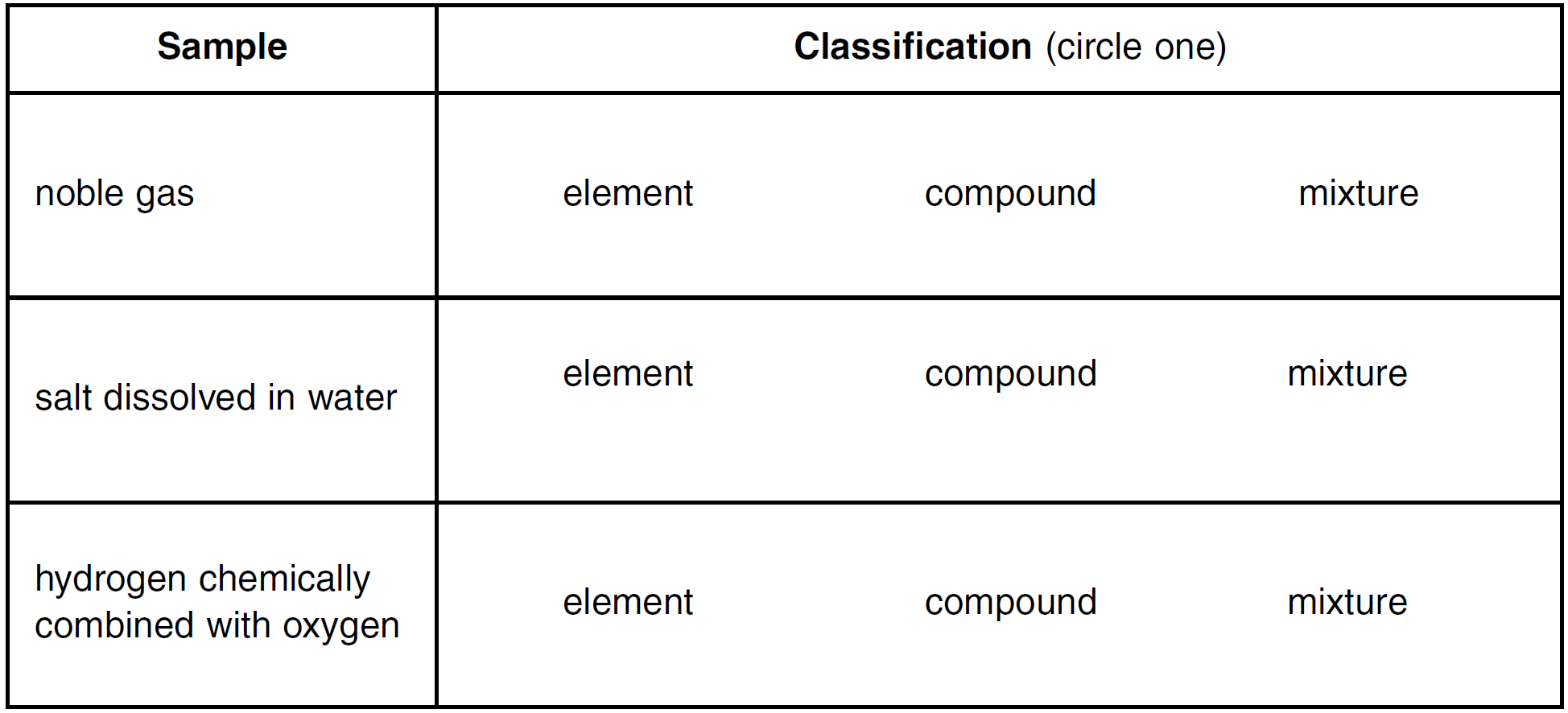
Noble gas: element
Salt dissolved in water: mixture
Hydrogen chemically combined with oxygen: compound
The diagram shows an overhead view of two trains, A and B, traveling beside each other in the direction shown by the arrow.
What visual evidence observed by the passengers in either train lets them know that their train is moving in the direction indicated by the arrow?
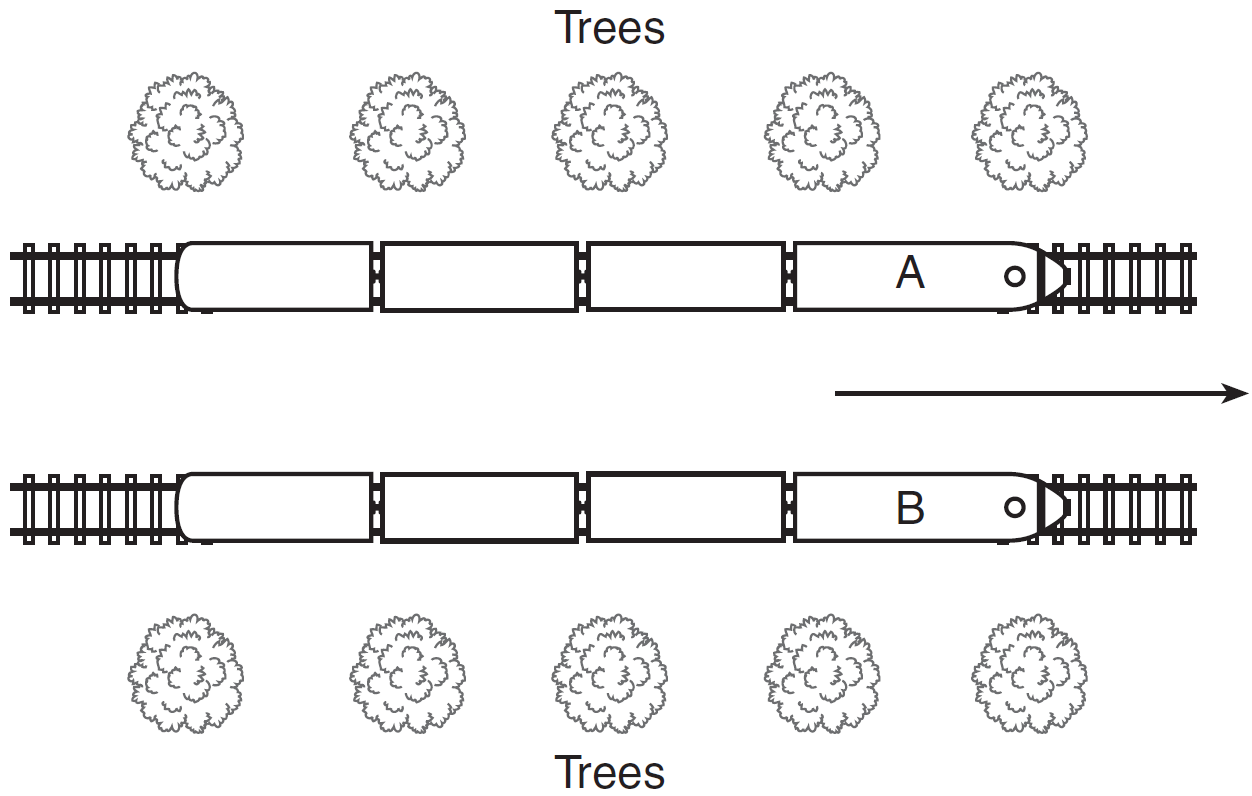
— When they look at any fixed object ahead of the train, it seems to approach, and then to move away in the opposite direction.
— The trees appear to move.
— The tracks appear to move.
— They use a reference point. For example, the trees appear to move but we know that the trees do not move, which shows that the train is moving.