What are the three main states of matter?
Solids, liquids, and gases.
In physical science, what are properties?
A property is any characteristic that is used to describe matter such as its color, material, or shape.
We use these characteristics to identify matter and distinguish them from one another.
For instance, that's how we can tell the difference between gold and silver.
A ____________________
contains two or more substances
that are not chemically combined.
Mixture
What is a chemical change?
A change that occurs when the particles that make up two or more substances are rearranged to form a new substance.
What is matter?
Anything that has weight and takes up space
This state of matter has a
definite shape and volume
and has particles that are
tightly packed together
Solid
Pick an object in this room.
What are some of its properties that can be observed?
Provide at least three.
Color, shape, material. You can tell just by looking at the object.
If matter can easily dissolve into a liquid
it has high _________________.
Solubility
What are some of the signs
that a chemical change is occurring?
Makes smells
Releases light
Releases gases
Gives off or takes in heat
What is an matter made of?
Tiny particles
___________________ happens when water vapor changes directly into ice —such a snowflakes and frost.
Notably, this is the process where water changes directly to ice without first becoming a liquid.
It usually happens at the top of mountains where temperatures are low.
Deposition (the opposite of sublimation)
Pick an object in this room.
What is one of its properties that can be measured?
Provide a measurement for that object.
Length, width, height, volume, weight. You need to use tools, numbers, and math in order to measure something.
Which type of solution has too much solute and no more solute will dissolve?
Saturation is the point at which no more solute can dissolve in a solution. For example, when you add too much sugar to water, at some point the sugar won’t dissolve anymore.
In a ___________ change,
a new substance is made,
like when you burn a candle.
In a ________ change,
no new substance is made,
like when water turns to ice.
In a chemical change,
a new substance is made,
like when you burn a candle.
In a physical change,
no new substance is made,
like when water turns to ice.
Provide three examples of matter in this classroom.
Everything is matter!!!
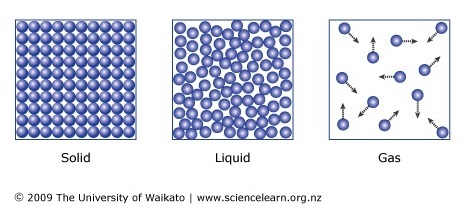
Come up to the board to draw a model that shows how the atoms behave in each of the three states of matter.
Which property of matter changes depending on the gravitational pull?
In other words, which property is affected by gravity?
Weight.
Imagine you are creating a solution. What is the solute in your experiment? What is the solvent in your experiment?
The solute is the substance that is dissolved in a solution. For example, sugar is a solute when mixed with water because it is dissolving in the water.
The solvent is the substance that does the dissolving in a solution. For example, water is the solvent when mixed with sugar because it is helping the sugar dissolve.
Provide an example of a chemical change and a physical change.
Nice :)
Provide an example of
each of the three states of matter
that cannot be found in this classroom.
Rad :)
Can states of matter change?
If yes, provide an example.
Yes they can!
How do you think
investigating properties of matter
might be useful in the real world?
It's important to tell the difference between substances that could be potentially harmful or unknown.
Sort between mixtures and solutions
using the examples provided.
Sweet! :)
Sort the following items between
physical and chemical changes.
Cool :)
What is the difference between an
atom and a molecule?
Atoms are tiny particles that are the basic building blocks of all matter.
Molecules, on the other hand, are made up of atoms that are held together by a chemical bond.