What three conditions must be satisfied for a chemical reaction to occur?
Collision, Orientation, and Energy
Reactants form products in the _____ reaction
Products form reactants in the FORWARD reaction
Products form reactants in the _____ reaction
Products form reactants in the REVERSE reaction
Which of the following bonds are polar?
H-C
O-O
N-H
F-C
F-O
N-H
F-C
F-O
What is the name of C2H3O2-?
Acetate
The name for the minimum amount of energy required to break the bonds between atoms of the reactants is what?
The activation energy (Ea)
In the following image, which of the following molecules is/are the product(s) ?
?
H2CO3
When does this reaction reach equillibrium?
14 minutes (accept 13-15)
Is this a polar or a non-polar molecule? If so, where is the negative dipole?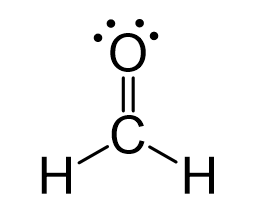
O = 3.5, H = 2.1, C = 2.5
Yes! The negative dipole is located on the O
What is the formula for hydroxide?
OH-
How dos a catalyst increase the rate of a chemical reaction?
It decrease the activation energy (Ea)
Write the equilibrium expression for the following chemical equation:
ClNO2(g) + NO(g) --> <-- NO2(g) + ClNO(g)
Kc = ([NO2][ClNO])/([ClNO2][NO])
Write the expression equilibrium expression for the following chemical equation:
Ca(OH)2 (s) + 2 H+(aq) --><-- Ca2+(aq) + 2 H2O(l)
Kc = [Ca2+]/[H+]2
Will CH4 dissolve into H2O?
No! (likes dissolves like)
What is the name of SO32-
Sulfate
Explain what would happen to the rate of a chemical reaction if I reduced the temperature of reactants and WHY this would be the case
The rate of reaction would slow as I would be decreasing the kinetic energy of the molecules meaning they would move slower, collide less often, and collide with less energy.
Consider the following chemical equation:
Al(OH)3(s) --><-- Al3+(aq) + 3OH-(aq)
What is the Kc for this reaction if, at equilibrium and -25 C, I have a concentration of 0.132 M of Al3+ and 0.342 M of OH- ?
Kc = 0.00528

Oh no!
Lose 400 points!
What intermolecular forces would be present in a cup of HNO2.
Dispersion forces
Dipole-Dipole
Hydrogen Bonding
Ion-Dipole
Covalent Bonding
Ionic Bonding
N = 3.0, O= 3.5, H = 2.1
Dispersion forces
Dipole-Dipole
Hydrogen Bonding
What is the formula for carbonate?
CO32-
On the following graph, identify where the products, the reactants, the activation energy, and the heat of the reaction are located. Then, determine if this is exothermic or endothermic
Exothermic
Consider the following reaction:
2SO2(g) + O2(g) --><-- 2SO3
800 K was maintained until the system reached equilibrium. The equilibrium mixture contained:
5.0 x 10-2 M SO3, 3.5 x 10-3 M O2, and 3.0 x 10-3 M SO2.
What is Keq for this reaction?
Keq = 7.9 x 104
Consider the following reaction:
H2(g) + I2(g) --><-- 2 HI(g)
740 K was maintained until the system reached equilibrium. The equilibrium mixture contained:
1.37 x 10-2 M HI, 6.47 x 10-3 M H2, and 5.94 x 10-4 M I2.
What is Keq for this reaction?
Keq = 48.8
Order the following from lowest to highest melting points.
CH3CH3
MgSO4
N2H4
KCl
C2H5F
Lowest
CH3CH3
C2H5F
N2H4
KCl
MgSO4
Highest
What is the name of NO3-
Nitrate

