What is the name of this isotope:

Carbon-14
What does EMR stand for?
Electromagnetic Radiation
What kind of radiation has the least penetrating power, and can be stopped by human skin?
Alpha radiation
After one half-life, how much of the parent isotope remains?
50%
When a large atom splits into smaller atoms, this is called nuclear _______.
Fission
Which element was named after water?
Hydrogen (it makes water when it reacts with Oxygen)
Isotopes are versions of the same element that differ in the number of ______ in their atoms.
neutrons
What is the most energetic light on the EM spectrum?
Gamma Rays
C-14 becoming N-14 is an example of what kind of decay?
Beta decay
If 87.5% of a daughter isotope has been produced, how many half-lives have passed?
3 half-lives
When a nuclear reaction releases multiple neutrons, which in turn trigger multiple additional reactions, this is called a ______ _______ (2 words).
chain reaction
Which element was named after the Sun?
Helium (after the Greek God Helios; the element was first discovered in the Sun!)
An isotope has 29 protons and 38 neutrons. What is the name of the isotope?
Copper-67
Ionizing radiation, which is dangerous and can cause mutations and cancer, has ___ energy, ___ frequency, and ___ wavelength.
HIGH energy, HIGH frequency, SHORT wavelength
An alpha particle is the same as the nucleus of which element?
Helium
Carbon-14 has a half-life of 5730 years. After 11460 years, how much Carbon-14 in a sample would remain?
25%
Fill in the blank:
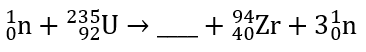

State an element named after a country (there are around 8 in total).
Francium, Polonium, Germanium, Nihonium, Ruthenium, Americium, Gallium, Indium
What is another name for Hydrogen-2?
Deuterium
How many nanometres are in one metre?
10^9 , one billion
Which type of decay has no charge?
Gamma decay
A radioactive substance has a half-life of 50 years. What fraction of the parent isotope remains after 500 years?
1/1024
Complete the equation.


State an element named after a planet (at least 3 or 4, depending on what you consider a planet!)
Mercury, Uranium, Neptunium, Plutonium
What is one reason why atomic masses are NOT always integers? (e.g. the mass of Magnesium is 24.3)
The atomic mass is an average, based on relative abundance of isotopes (e.g. Magnesium on Earth is a mix of Magnesium-24, Magnesium-25, and Magnesium-26)
A certain radio wave has a wavelength of 4 km, what is its frequency?
75000 Hz, or 75 kHz
If Uranium-236 undergoes alpha decay, what daughter isotope is produced?
Thorium-232
A radioactive substance has a half-life of 1000 years. What % of the substance remains after 500 years? (round to nearest %)
71%
What is another name for Hydrogen-3?
Tritium
State an element named after a woman (2).
Curium, Meitnerium