What are the two reactants in any hydrocarbon combustion reaction?
fuel and oxygen
Does the following PE diagram show an exothermic or endothermic reaction? How do you know?
exothermic, PE products is lower than PE reactants
How do we represent IMFs when drawing them?
Use dotted lines
What is Avogadro's number?
6.02 x 1023
What is the name of the leading number in front of a chemical compound in a reaction (ex: the 2s in 2H2 + O2 --> 2H2O)?
coefficient
What are the two parts of every solution?
solute and solvent
What are the two products in any hydrocarbon combustion reaction?
CO2 and H2O
Which letter on the PE diagram represents the activation energy of the forward reaction?
B
What is the four letter acronym used to memorize molecular polarity trends?
SNAP
What is the gram formula mass (molar mass) of C2H4?
28 g/mol
What is the mole ratio between N2 and NH3 in the Haber reaction below?
N2 + 3H2 --> 2NH3
1:2
What is the maximum mass of sucrose that can be dissolved in 100 mL of water at 60oC?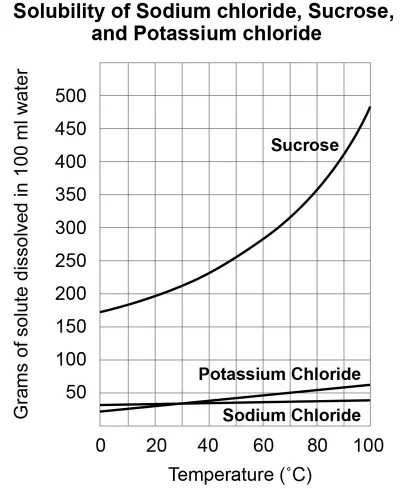
Which of the following describes the relationship between bond stability and reactivity?
a. the more stable the bond, the less reactive the compound
b. the more stable the bond, the more stable the compound
c. both a and b are correct
d. neither a nor b are correct
B
What PE value corresponds to the activated complex in the following diagram?
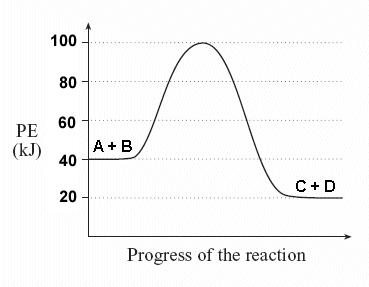
100 kJ
Describe the strength of the IMFs in ethane as shown below.
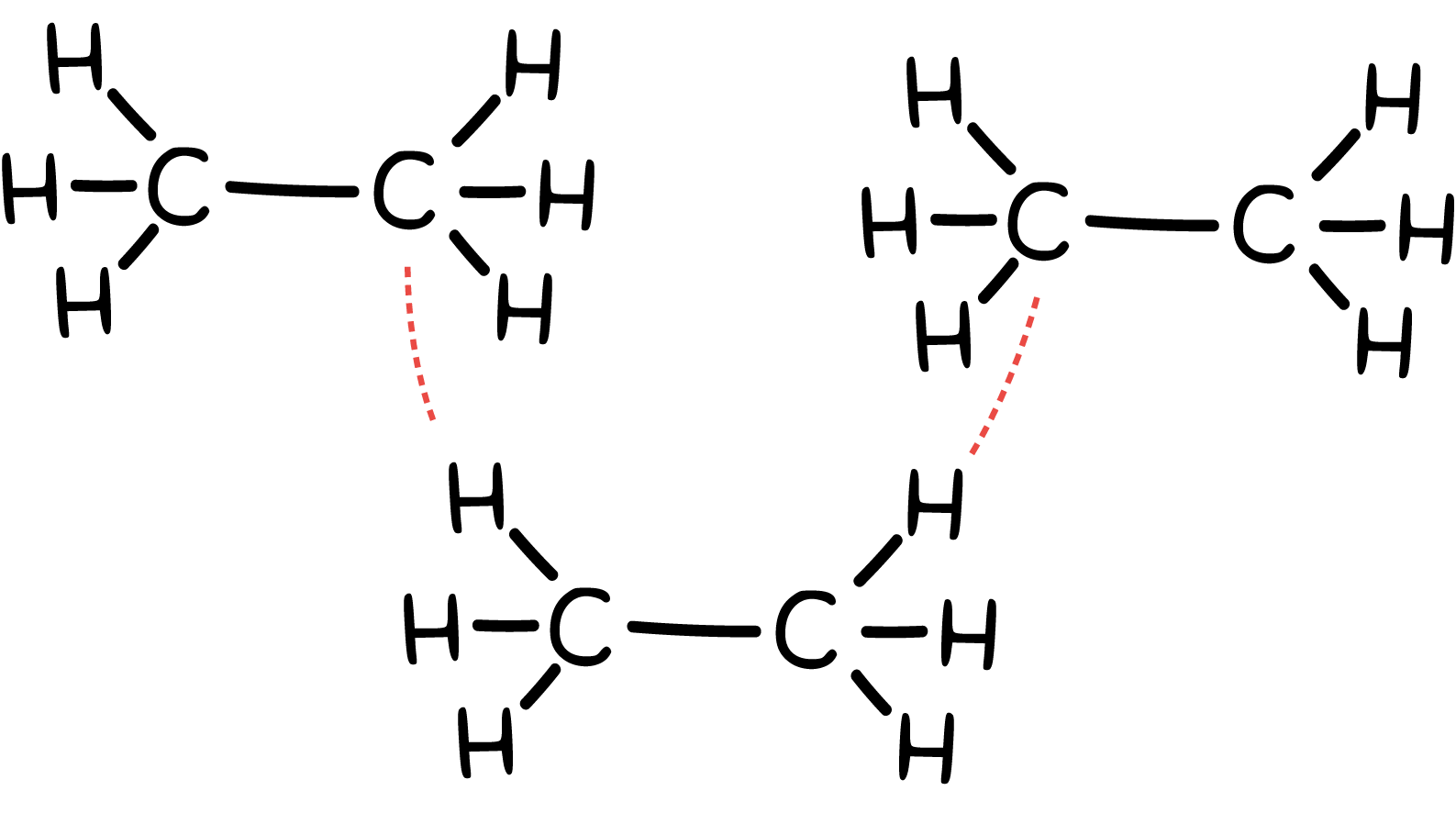
very weak
Convert 12 g of water to moles of water. Round to the nearest hundredth place.
0.67 moles
How many moles of ammonia (NH3) are produced when 4.2 moles of H2 react with excess N2?
N2 + 3H2 --> 2NH3
2.8 moles of NH3
What is the molarity of an NaCl (aq) solution when 58 g of NaCl is dissolved in a 0.5 L solution?
2 M
Which of the following correctly shows the relationship between bond length, bond energy, and bond strength?
a. the greater the bond length, the greater the bond energy, and the greater the bond strength
b. the greater the bond length, the lesser the bond energy, and the greater the bond strength
c. the greater the bond length, the lesser the bond energy, and the lesser the bond strength
d. none of the above
C
What is the heat of reaction
(DeltaH)?
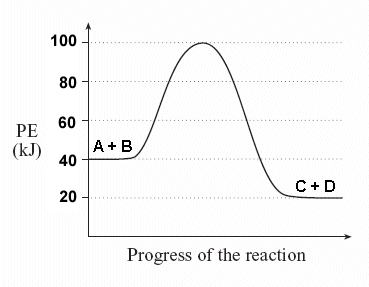
-20 kJ
In hydrofluoric acid, HF, which side would get the delta - and which side would get the delta +
H would be
delta+ and F would be
delta-
Convert 3.40 moles of NaCl to grams of NaCl. Round to the nearest whole number.
197 g
2 NaCl → 2 Na + Cl2
How many grams of sodium chloride (NaCl) are required to make 0.32 moles of chlorine (Cl2)?
37 g NaCl
What is the concentration in parts per million of an AgCl(aq) solution that contains 1.2 x 10-3 grams of AgCl in 800 grams of the solution?
1.50 ppm
Based on the bond energies provided, list the covalent bond pairings from least stable to most stable?
C-N, C-C, C-O, C-H
The difference of which two letters would result in the activation energy of the reverse reaction? (ex: X - Y)

C - D
What is the name of the IMF in red, and what is the relative strength of this IMF?
hydrogen bonding, strong IMF
How many molecules of water are in 230 g of water?
7.7 x 1024 molecules
2 Ga2O3 → 4 Ga + 3 O2
How many grams of oxygen (O2) can be produced from 15.2 grams of gallium oxide (Ga2O3)?
3.88 g
If 25.0 mL of a 2.19 M solution are diluted to 72.8 mL, what is the final concentration?
0.752 M