What is the preferred ion charge for Sodium?
1+
What family of elements does Calcium belong to?
Akali Earth Metals
An ionic compound is made of what kinds of elements?
A metal & a non-metal
A molecular compound is a combination of what kinds of elements?
2 Non-metals
If you tasted a sample of acetic acid, how would you expect it to taste?
Sour
What is the name for a positive ion?
Cation
82
What is the name for the following compound: SrF2?
Strontium Fluoride
What is the name of the following compound: CBr4
Carbon Tetrabromide
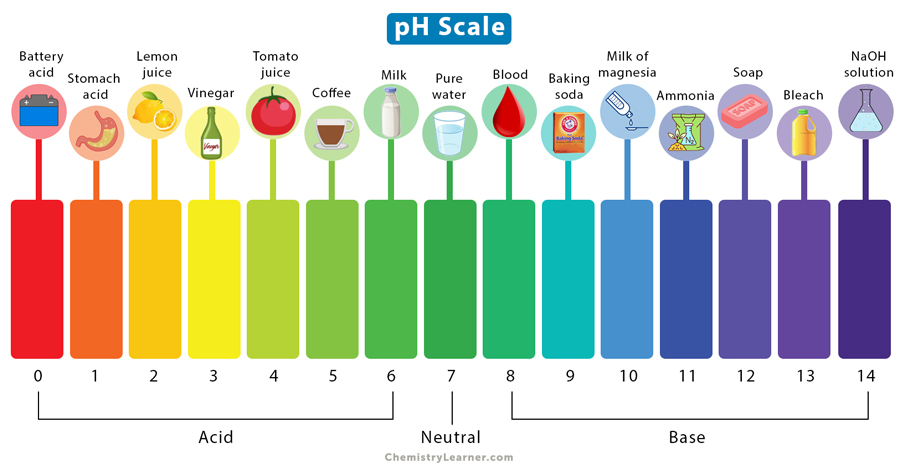
How much stronger of an acid is Lemon juice compared to Tomato juice?
100x
What is the name for the following ion: Cr2O72-(aq)?
Dichromate ion
You have a sample of a dull, yellow element that does not conduct electricity. What class of element do you think it is?
How does the melting point of ionic compounds compare to molecular compounds?
Ionic is higher
The electrons of molecular compounds are _________ between each atom?
Shared
An unknown sample turned all litmus paper blue. What kind of compound is it?
A base
What is the term for elements with multiple possible ion charges?
Multivalent
How many neutrons does Thorium have?
142
What is the chemical formula for Indium Oxide?
In2O3
What is the chemical formula for the following compound: Tetraphosphorous Decaoxide?
P4O10
The concentration of H+(aq) does what when pH decreases?
Concentration increases
Which of the following elements has the same number of electrons as a chloride ion (Cl1-)?
Argon atom
Which metal is the only one that is liquid at room temperature? (Hint: It's toxic)
Mercury
You have an sample on an unknown ionic compound and place it in water. What do you expect to happen?
It will dissolve
You have a sample of carbon tetrachloride. What would happen if a conductivity meter was placed into the sample?
It would not conduct
What is the name for the H2CO3(aq) dissolved in water?
Carbonic acid