What type of heat transfer occurs through direct particle contact?
Conduction
What force holds oppositely charged ions together in an ionic bond?
Electrostatic force
What happens to atomic radius across a period from left to right?
Atomic radius decreases.
Which subatomic particle determines an element’s identity?
Protons
What charge does sodium form when it becomes an ion?
+1
Complete this sentence about convection currents: Hot fluid is _____ dense and _______; cool fluid is ______ dense and ______.
Hot fluid is less dense and rises; cool fluid is more dense and sinks.
Which is stronger? A covalent bond or an ionic bond? Why?
Ionic bonds due to electrostatic force.
Why do elements in the same group have similar properties?
They have the same number of valence electrons.
What happens to its charge when an oxygen atom gains two electrons?
O2- ion
How can you identify an ionic compound by its elements?
Ionic compounds contain a metal and a nonmetal.
A calorimeter is a device that measures heat changes and is insulated. That means that the total thermal energy in the calorimeter...
(Increases/Decreases/Remains the same/)
The total thermal energy remains the same.
N2 + O2 --> 2NO (and is formed during electrical storms).
Which bonds are broken in this reaction?
Bonds between nitrogen and oxygen
Which reacts more vigorously with water, calcium or magnesium?
Calcium, because it has a larger atomic radius and lower ionization energy. (easier to remove electrons)
What do the dots in a Lewis dot diagram represent?
Valence electrons
What is the charge of iron in Fe₂S₃
3+
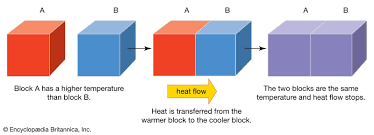 What happens to blocks A and B after heat flow stops? Temperatures --> A is greater than B, B is greater than A, both blocks reach the same temperature
What happens to blocks A and B after heat flow stops? Temperatures --> A is greater than B, B is greater than A, both blocks reach the same temperature
Both blocks reach the same temperature
Name the two compounds:
Fe3(PO4)
C2H4
Dicarbon tetrahydride
Why does ionization energy decrease down a group? (Use shielding or nuclear attraction)
Increased electron shielding makes valence electrons easier to remove.
What does an atomic emission spectrum show about electrons?
Electrons exist in specific energy levels.
What is the charge of OH in Pb(OH)2
-1
What heat transfer process drives movement in the mantle? Where is the source of its heat?

Convection currents from the inner core
Which will have a greater melting point?
Na3PO4 or C2H4
Na3PO4
Order the following from smallest atomic radius to largest
Rb, Li, K, Na, Cs
Li, Na, K, Rb, Cs
What happens when an electron drops from a higher energy level to a lower energy level?
It emits energy in the form of heat and light
If an compound is not conductive as a solid, but is conductive when dissolved in water, it most likely is... (ionic/covalent)
Ionic