2-6 is the electron configuration for which element?
Oxygen
Ammonia
Name the following Acid:
HNO2
Nitrous Acid
How much is 1 mole worth?
6.02x1023
 Name a section where potential energy is decreasing
Name a section where potential energy is decreasing
Condensing or Freezing
How could you change the configuration 2-3 to show Boron's electrons in an excited state
2-2-1
HIGH Temperature & LOW pressure
Which of the following is INSOLUBLE in water
a. NaCl
b.CaCrO4
c. SrSO4
d. (NH4)2CO3
Strontium Sulfate
SrSO4
Calculate the mass of a 5 mole sample of Chloromethane: CH3Cl
C;12 H; 1x3 Cl 35.5 x 1 50g
5 mol x 50g / mol = 250g
This theory states that all particles are in motion
Kinetic Molecular Theory
Transition metals are part of which orbital group:
a.s
b.p
c.d
d.f
D Block
Define: Entropy
"A lot of kids' rooms exemplify entropy."
a. HCl
b. NH3
c. NH3Cl
d. KClO3
A. HCl
A 12g sample of water increases in temperature from 283K to 305K, calculate the amount of energy added.
q=mCAT = (12)(4.18)(22) = 1103.52J
q=?
m=12g
C=4.18 J/g*K
Delta T= 305-283 = 22
Put the Atomic Theorists in order
Schrodinger, Dalton, Bohr, Rutherford, Democratus, Thomson
Democratus, Dalton, Thomson, Rutherford, Bohr, Schrodinger
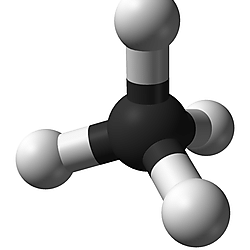 What molecular structure does Methane have?
What molecular structure does Methane have?
Tetrahedral
According to the Colligative Properties, pure water will have a (HIGHER / LOWER) freezing point than salt water.
Higher
Salt water has a lower freezing point
Would the following single replacement reaction Occur to produce Hydrogen Gas? (Yes or No)
2HCl + Mg --> MgCl2 + H2
Yes! Use Table J
Calculate the change in pressure of a 51mL sample gas currently at STP when the Temperature is increased to 341K and Volume has changed to 25mL
P1V1/T1 = P2V2/T2
P2 = (1atm)(51mL)(341K) / (273K)(25K)
P2 = 2.5atm
Compare electron movement in Ionic and Covalent Bonds
Ionic bonds have taken/given electrons and covalent bonds have shared electrons.
Substances that evaporate slowly have ___________________ intermolecular forces.
(stronger / weaker)
Stronger

In the FORWARD reaction, which of the following would be considered the CONJUGATE Base
OH-
Hydroxide
a. Rubidium
b. Argon
c. Chlorine
d. Barium
a. Rubidium! (215)
b.101
c.100
d.206
Calculate the initial Concentration of an Acid if I combine a 37mL sample with a 41mL 1.2M Sample of a base.
MAVA = MBVB
MA = (41mL)(1.2M) / (37ml)
MA = 1.3M