Define solute
What is being dissolved by the solvent (you add it)
What does M stand for in chemistry?
Molarity
What happens to concentration when you add more solvent?
A: It decreases
What does a solubility curve show?
How much solute dissolves at different temperatures
In salt water what is the solute and what is the solvent
salt= solute , Water=solvent
Define a solvent
The part of a solution that does the dissolving
How many moles are in 1 L of a 2.0 M solution?
2.0 moles
If you dilute a solution from 1.0 M to 0.5 M, what happens to volume?
It doubles
If a point is on the curve, what type of solution is it?
Saturated
How do you do well on tests and quizzes
study
A solution that is below its maximum capacity for a solute is identified as
unsaturated
What is the molarity if 0.5 moles are dissolved in 2 L?
0.25 M
: A 1.0 M solution becomes 0.25 M. By what factor did the volume change?
It increased by 4 times
If a point is below the line the solution is considered
unsaturated
initial concentration (M1): 2.4 M
initial volume (V1): 4600 mL
final concentration (M2): 2.168 M
What is the new volume
5.1L
What term describes a solution that contains the maximum amount of solute?
Saturated
How many moles are in 0.75 L of a 3.0 M solution?
2.25 moles
You have 2.0 M and want 1.0 M. If you start with 100 mL, what is final volume?
200 mL
If a point is above the curve, what type of solution is it?
A: Supersaturated
Temperature at which you can dissolve 80g of sodium nitrate
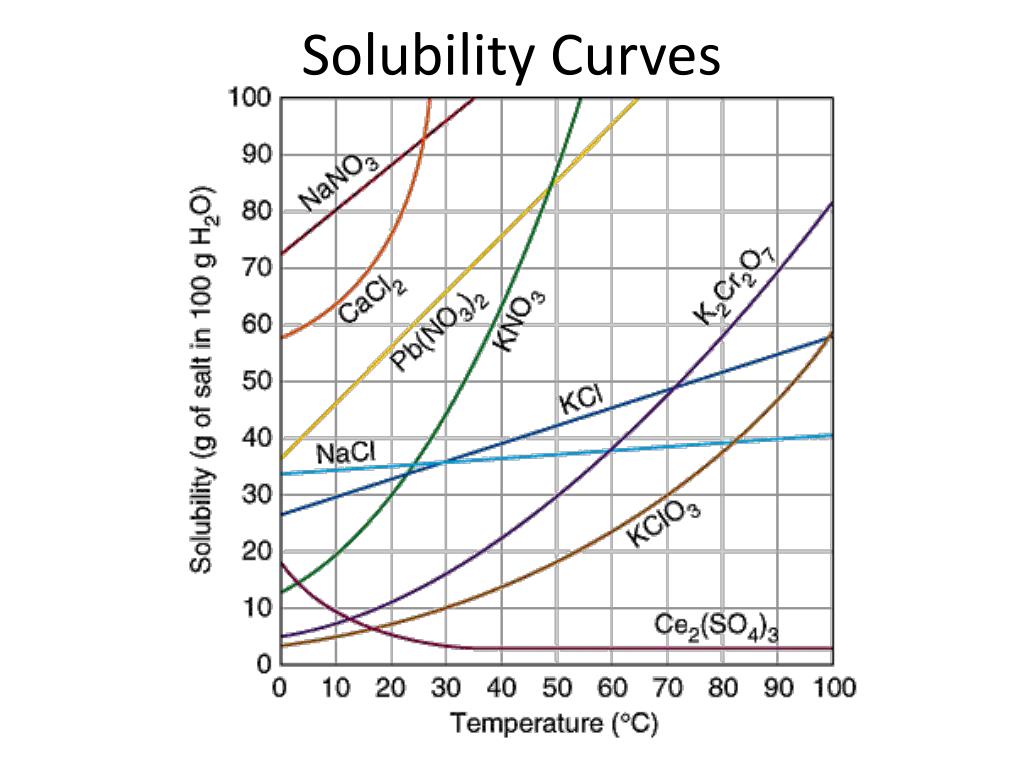
10 Celsius
Describe how one gets a super saturated solution
heat--> dissolve more--> cool down
if 10g of NaCl is dissolved in 100ml of water what is the molarity
1.71M
A 150ml solution 0.75M is diluted to 1/3 of its concentration what volume would the new solution be in Liters
0.450L
What generally happens to solubility of solids as temperature increases?
A: It increases
initial concentration (M1): 1.47 M
initial volume (V1): 3300 mL
final volume (V2): 4100 mL
How many moles of solute would be present
4.85 moles