What are the electrons in the outer energy level called?
Valence electrons
What is the part of an atom with a negative charge?
Electron
What information does the atomic number provide?
The number of protons (and electrons) an atom of an element has.
What is the formula for finding the neutrons of an atom?
The atomic mass minus the atomic number
What is a subatomic particle with no charge?
Neutron
Where are protons and neutrons located?
Inside the nucleus?
The group of atoms with a completely full outer most (valence) energy level?
Noble Gases
What are the tiny particles that make up all matter?
Atoms
How many protons and electrons does Arsenic (As) have?
33
The location of electrons.
What is outside the nucleus?
The columns on a periodic table are called?
Groups
What is the maximum number of electrons the 2nd orbital, or energy shell, can hold?
Eight.
What do we call the rows on the periodic table?
Periods
How many neutrons does Arsenic (As) have?
42
Elements in the same group share what?
The same number of valence electrons
What is the maximum number of electrons the 1st orbital, or energy shell, can hold.
Two
What is the part of an atom with a positive charge?
Proton
The majority of the elements on the periodic table are what type?
Metals
What is the distance from the center of the atom's nucleus to the electrons in its outermost energy level?
Atomic radius
The three classes of elements are these.
What are metals, metalloids, and nonmetals?
What elements are found in a zigzag line on the periodic table?
Metalloids
How is the modern periodic table is organized?
By increasing atomic number
Which group of elements are considered extremely reactive?
Alkali Metals
This representation of an atom with its valence electrons is called what?
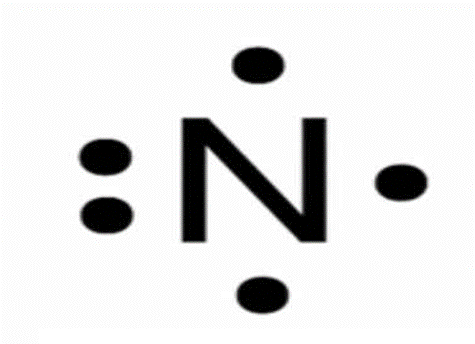
The electron dot notation
An element's ability to attract and hold electrons when bonded to other atoms is called what?
Electronegativity