Temperature
When a thing gets hotter, its molecules are moving...
Faster

Soup of the same temperature was placed in both bowls. Which one had more thermal energy?
Bigger bowl because it had more molecules
Explain the total and average KE of a very small, but very hot flame
High average KE and low total KE
Give an example where a substance has low average KE and high thermal energy (total KE)
Anything large and cooler in temp
The total amount of energy in a substance
Thermal energy
When a thing gets colder, its molecules are moving...
slower
Find the average of these numbers:
3, 5, 7, 10, 5
6
Which has more thermal energy: a large block of ice or a small cup of warm coffee?
Large block of ice
Give an example of a substance with high average KE and high total KE
A giant fire (large and hot)
The moment when two objects hit each other
Collision
Temperature is a measure of the ____________ kinetic energy.
Average
Find the total kinetic energy is each the following numbers represented the KE in each molecule:
4, 2, 8, 6, 10
30
Increase
Give an example of a substance with low average KE and low thermal energy
Small piece of ice (something small and cool)
A balanced state in which a system is stable
Equilibrium
When a thing gains of loses energy, the energy gained/lost is divided among ______ the molecules of a thing
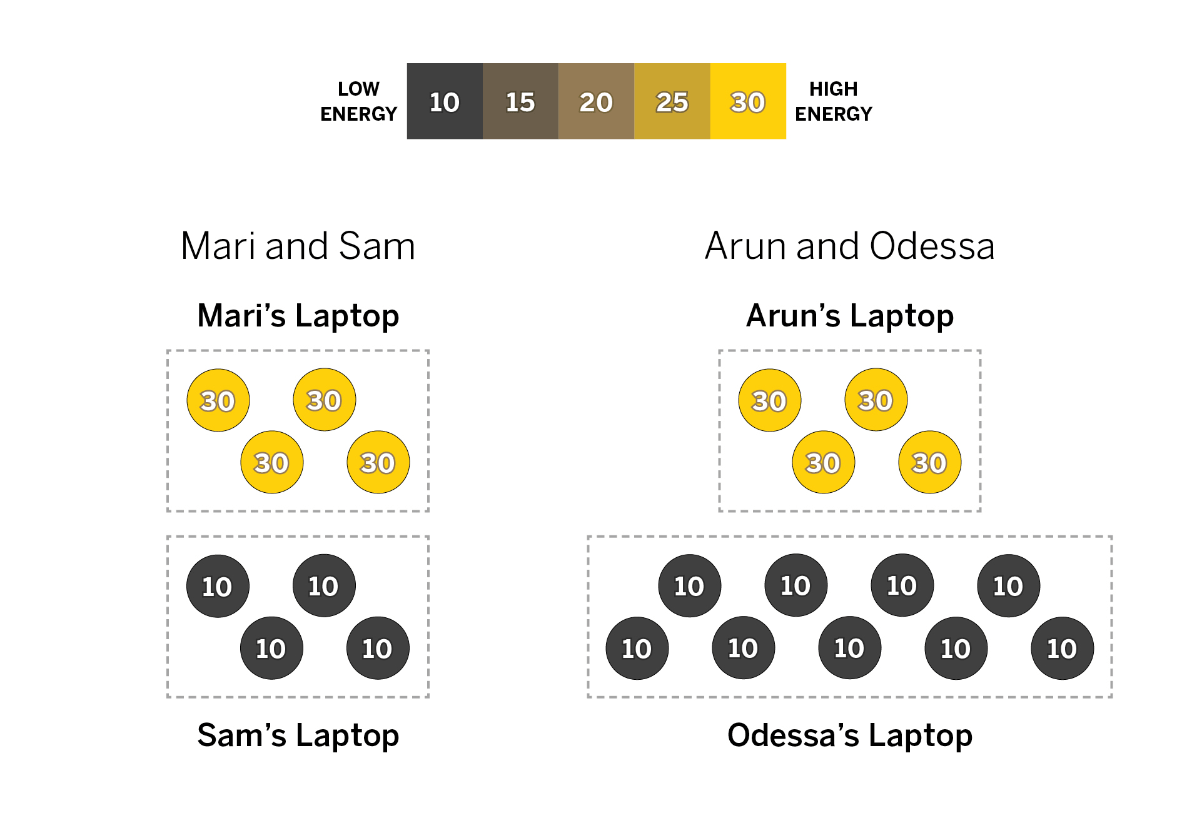
What is the equilibrium average KE of Sam and Mari's laptop?
20
 Calculate the average KE after the two substances reach equilibrium
Calculate the average KE after the two substances reach equilibrium
26.15
Give an example of a substance that has high average KE and low total KE
Small and hot (a firework)
A measure of how hot or cold something is
When two things reach equilibrium, they will have the same...
Temperature or average kinetic energy

Which set of laptops will have a higher average KE when equilibrium is reached? (no calculation needed here)
Odessa and Arun

What is the average KE of Odessa and Arun's laptop when they reach equilibrium?
~16
16.15
 What is wrong with this model at equilibrium?
What is wrong with this model at equilibrium?
Not all molecules have the same KE