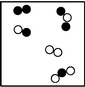
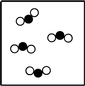
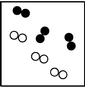
What is the smallest unit that defines an element?
A. Element
B. Plasma
C. Compound
D. Atom
D. Atom
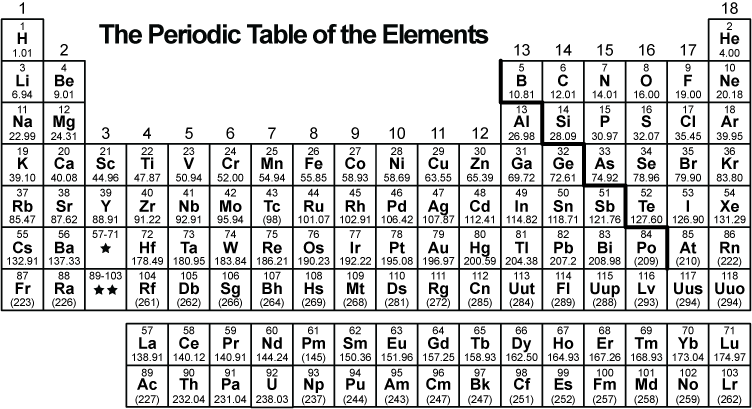
This is the periodic table of the elements.
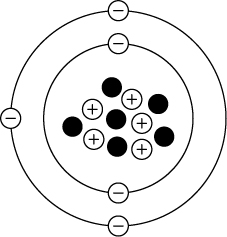
The diagram shows an unknown atom.
Based on the information from the periodic table, which atom is shown in the diagram?
A. boron
B. carbon
C. nitrogen
D. Oxygen
The periodic table is organized into vertical groups of
A. compounds with different properties.
B elements with similar properties.
C. elements with all the same chemical properties.
D. compounds with all different physical properties.
elements with similar properties.
Pure substances are:
A.elements and can be represented by one chemical symbol.
B. alloys and can be represented by one chemical symbol
C. mixtures and can be represented by two or more chemical symbols.
D. solutions and can be represented by two or more chemical symbols
elements and can be represented by one chemical symbol.
In science lab, Ellie combined sugar and distilled water. Ellie made
A. a mixture, which must be separated by chemical processes.
B. a compound, which must be separated by chemical processes.
C. a mixture, which can be separated through physical processes.
D.
a compound, which can be separated through physical processes.
a mixture, which can be separated through physical processes
What is formed when two or more elements are combined chemically?
A. mixture
B. ion
C. nucleus
D. Compound
D. Compound
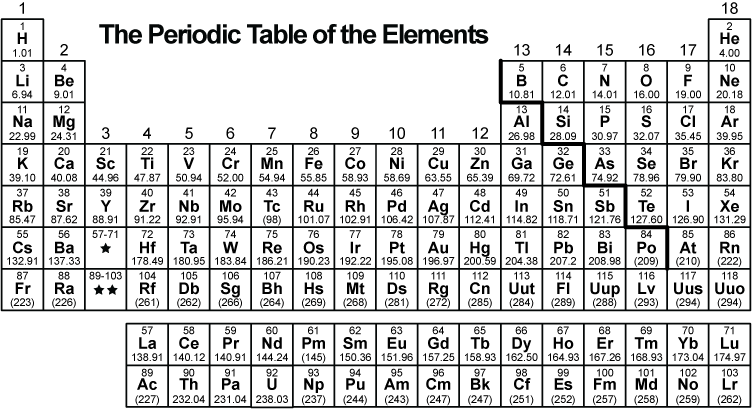
The illustration below, taken from the periodic table, provides information about the element calcium (Ca).
Where would the element with atomic number 19 be placed on the periodic table?
A. above calcium
B. below calcium
C. to the right of calcium
D. to the left of calcium
to the left of calcium
Two particles of the element iodine are chemically bound. The two particles compose what?
A. an atom
B. a compound
C. a molecule
D. A mixture
a molecule
Air contains about 78% nitrogen and 21% oxygen. Based on this information, one can conclude that air
A. is a pure substance.
B. consists of components that are chemically joined.
C. consists of different states of matter.
D. is a mixture of at least two gases.
is a mixture of at least two gases.
Marcy creates a model that compares elements and compounds. Her model is shown.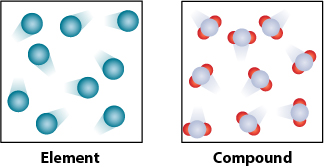
A. An element is highly reactive, and a compound is stable.
B. An element can exist in any state, and a compound can only be liquid.
C. An element contains multiple substances, and a compound contains only one pure substance.
D. An element is made of a single type of atom, and a compound is made of two or more atom types.
An element is made of a single type of atom, and a compound is made of two or more atom types.
How many atoms of chlorine are in a molecule of phosphorus trichloride, PCl3?
A. 1
B. 2
C. 3
D. 4
3
Aluminum sulfate has the following chemical formula:
Al2(SO4)3
How many different elements are found in aluminum sulfate?
A. 4
B. 3
C.5
D. 6
3
On the periodic table, atomic mass
A. increases across a period from left to right.
Atomic mass increases across a period from left to right.
B. decreases down a group.
Atomic mass decreases up, not down, a group.
C. decreases across a period from left to right. Atomic mass decreases across a period from right to left, not from left to right.
D. increases up a group. Atomic mass increases down, not up, a group.
increases across a period from left to right.
Atomic mass increases across a period from left to right.
Select all that are compounds.
A. I
B. KBr
C. NaK
D. NaCl
E. W
B. KBr
C. NaK
D. NaCl
Suzy is examining a brass lamp. If brass is a mixture of copper and tin, what can Suzy conclude?
A. Brass is an impure substance composed of copper, Cu, and tin, Sn.
B.Brass is a pure substance represented by the symbol B
C. Brass is a pure substance represented by the symbol Br.
D. Brass is an impure substance composed of copper, Co, and tin, Tn
Brass is an impure substance composed of copper, Cu, and tin, Sn.
This is the periodic table of the elements.
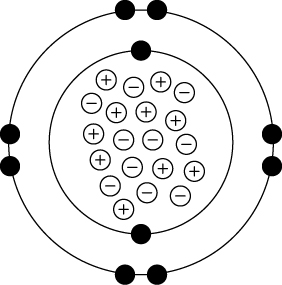
Using the periodic table, Maria drew a diagram of a neon atom. The diagram shows what Maria drew.
Which mistake did Maria make on her diagram?
A. The protons should be circling the nucleus, not the neutrons.
B. The neutrons should be circling the nucleus, not the protons.
C. The nucleus should have electrons and neutrons, not neutrons and protons.
D. The nucleus should have protons and neutrons, not protons and electrons.
The nucleus should have protons and neutrons, not protons and electrons.
Sam's mother is cooking breakfast in a cast-iron frying pan. If the pan is made only of iron, what can Sam conclude?
A. The pan is a pure substance with the chemical symbol Fe.
B. The pan is a pure substance with the chemical symbol F.
C. The pan is a pure substance with the chemical symbol Ir.
D. The pan is a pure substance with the chemical symbol I.
The pan is a pure substance with the chemical symbol Fe.
Substances X, Y, and Z need to be identified. Substance X can be broken down by a physical change. Substance Y cannot be broken down by a chemical or physical change. Substance Z can only be broken down by a chemical change. What can be concluded about these substances?
A. Substance X is a compound, substance Y is a mixture, and substance Z is an element.
B. Substance X is an element, substance Y is a compound, and substance Z is a mixture.
C. Substance X is a mixture, substance Y is an element, and substance Z is a compound.
D. Substance X is a compound, substance Y is an element, and substance Z is a mixture.
Substance X is a mixture, substance Y is an element, and substance Z is a compound.
Which particle diagram represents a pure substance?
A.
B.
C.
D.