What is an atom?
The smallest unit of matter.
24
What is an ion?
A charged atom. An atoms that's gained or lost electrons.
What is an isotope?
Atoms with the same number of protons but different number of neutrons.
What direction does a period go?
Horizontal (left to right)
Where can you find the atomic number? Is it the larger or smaller number.
Periodic table, it is the smaller of the two numbers.
What seperates the metals from nonmetals?
Metalloids (Stairstep)
What are the two types of ions? What are their charges?
Cation-positive and anion-negative.
There are two ways to notate an isotopes. Show both notations for carbon-13.
C-13 or 13C
Calculate the number of neutrons for thw two isotopes?
17O-2 16O
9 and 8
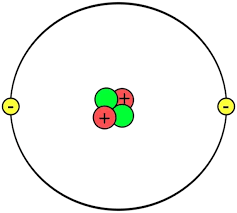 Given the following image, what element is this?
Given the following image, what element is this?
Helium
How many neutrons does the element with 52 protons have?
76 neutrons
P=3
N=4
E=2
Write element name and correct symbol.
Lithium. Li+1
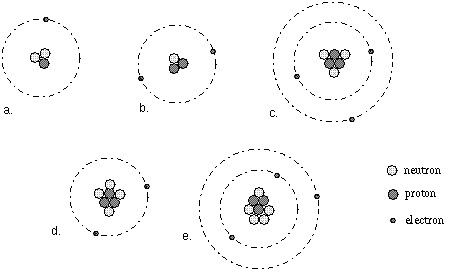
Which of the following are isotopes of one another and why?
C, D and E are isotopes because all of them have the same number of protons (3). They are all isotopes of Lithium.
Locate the element that contains 12 protons with an atomic number of 15
Cannot be possible/determined
What is the charge of an atom? What has to be true to make this happen?
Atoms have zero charge (neutral). Protons must equal electrons.
What subatomic particles are located in the nucleus?
Protons and neutrons
An element has 15 protons and 18 electron, is this an ion? If so, what type? Correct symbol?
Ion. Anion. P-3
 Which of the following are isotopes and why?
Which of the following are isotopes and why?
B and C are isotopes. They are Lithium isotopes. They have the same protons but different neutrons.
Bonus: Which of the following are ions?
A scientist has a Carbon atom. The scientist moves 4 electrons to the anion. How many protons and electrons does Carbon have?
6 protons, 8 electrons
What is the difference between the atomic mass and mass number? Where are they located?
Atomic mass is the average mass of the isotopes found on the periodic table. This is the mass of an atom. Mass number is the mass of the specific isotope. It is not found on the table but given.
Ex: X-m format
What is special about Mercury and Bromine?
They are metals but liquid at room tempurtature
e= 28 p=29
p=29 e=27
Given the information above, are these the same element? If so, what element? What are their correction symbols/notation?
Yes; they are both copper. Cu2+ and Cu+1.
Calculate the number of neutrons in Bi-179
96
What is the difference between average atomic mass and mass number?
Average atmoic mass is the average of all isotopes and mass number is the sum of protons and neutrons.