These are used in naming covalent compounds and indicate "how many" of an atom is present.
What is a prefix?
The formula formula for carbon tetrahydride is __.
Whate is CH4?
This is the type(s) of element(s) that most often participate in covalent bonding.
What is a nonmetal?
Draw the electron dot diagram for the element aluminum.
What is

The number of covalent bonds that hydrogen usually forms.
What is one covalent bond.
The name for the compound N2O5 would be_.
What is dinitrogen pentoxide?
The formula for iodine pentafluoride is__.
What is IF5?
Atoms that participate in covalent bonding do this with electrons.
What is share or sharing?
Draw the electron dot diagram for the element phosphorus.
What is

The number of covalent bonds that carbon usually forms.
What is four covalent bonds?
The name for the compound CF4 would be_.
What is carbon tetrafluoride?
The formula for tetraphosphorus trisulfide is_.
What is P4S3?
This is a neutral group of atoms held together by covalent bonds.
What is a molecule?
A single bond contains this many electrons.
What are 2 electrons?
This element is always in the center of a Lewis structure.
What is carbon?
Rule: The least electronegative element is in the center of a Lewis structure (except for hydrogen)
The name for the compound S3Cl6 would be_.
What is trisulfur hexachloride?
The formula for the compound dichlorine heptaoxide is_.
Cl2O7
Two identical elements bonding together are called....
What is a diatomic element or a diatomic molecule?
A triple bond contains this many electrons.
What is 3 electrons?
This is a pair of electrons that does not participate in bonding.
What is a lone pair?
The name for the compound Cl2O would be_.
What is dichlorine monoxide?
The formula for the compound disulfur decafluoride is_.
S2F10
The seven diatomic twins are_. (list)
What are Br, I, N, Cl, H, O, and F
This word is used to describe a molecule in which double bonds can move from one location to another.
What is resonance?
There is something wrong with this lewis structure.
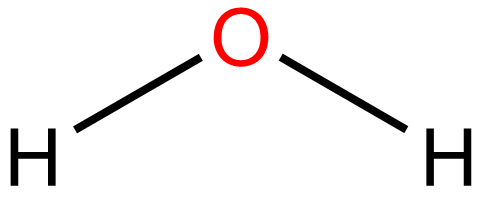
What is "no lone pairs on oxygen atoms"?
The name for the compound ICl would be_.
What is iodine monochloride?
The name below cannot be correct because of this rule_.
monophosphorus pentachloride
What is "never use the prefix mono for the first element in the name of a covalent compound"?
The seven diatomic elements all exist in this state of matter.
What is a gas?
A molecule that has an overall positive or negative charge is called this.
What is a polyatomic ion?
This element is never in the center of a Lewis structure.
What is hydrogen?