Identify the type of reaction:
S8 + 8O2 -> 8SO2
Combination
Ca + CuF2 -->
Ca + CuF2 --> CaF2 + Cu
Li2O -->
2Li2O -> 4Li + O2
Describe a way in which you could increase the rate of a chemical reaction:
Answers may vary:
1) increase surface area put breaking apart a solid reactant
2) increase concentration of reactants
3) increase the temperature of the reactants
4) add a catalyst to the reaction
What is the first condition necessary for a chemical reaction?
the reactant particles must collide.
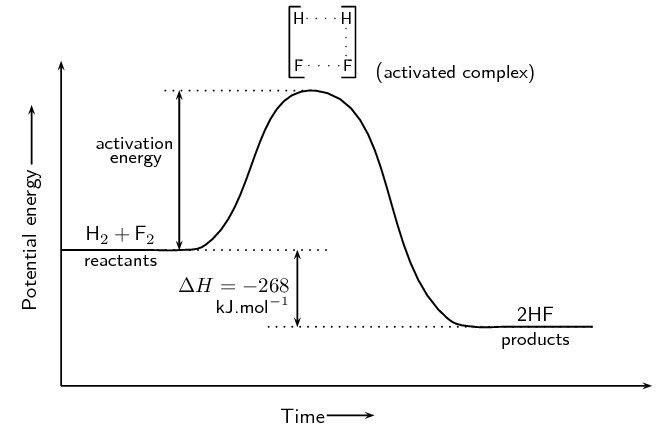
Identify the type of reaction:
2HF -> H2 + F2
DAILY DOUBLE!: What type of molecule does the reaction produce?
Decomposition
Diatomic molecules
Rb + O2 -->
4Rb + O2 -> 2Rb2O
H2SO4 + NaOH -->
H2SO4 + 2NaOH -> Na2SO4 + 2H2O
How does concentration affect the rate of a chemical reaction?
When the concentration is increased, there are more particles of reactant present in a given volume. This means that the chances of the reactant particles colliding to form new products is higher, because there are more particles present. This would therefore INCREASE the rate of the reaction.
DAILY DOUBLE!
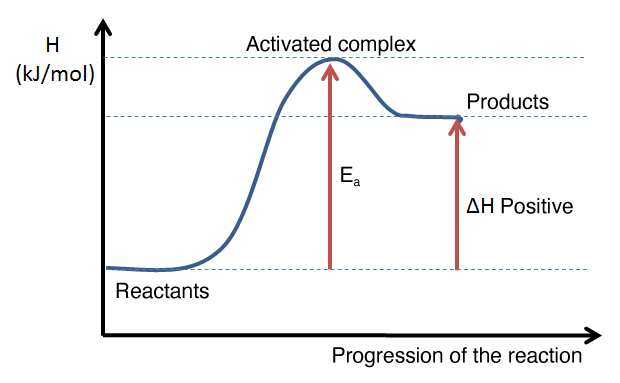
What is activation energy? What is the activated complex?
Draw an accurate energy diagram indicating the activation energy and the activated complex.
Ea = the minimal energy needed to produce a chemical reaction (energy needed to break apart the bonds of the reactants)
Activated Complex: intermediate step of a reaction in which the reactant bonds are broken, and the new bonds of the products are starting to form. It is the highest energy state of a chemical reaction.
Identify the type of reaction:
HI + CsOH -> CsI + H2O
Neutralization
HF + Ca(OH)2 -->
2HF + Ca(OH)2 -> CaF2 + 2H2O
Cl2 + PbI4 -->
2Cl2 + PbI4 -> PbCl4 + 2I2
Which factor(s) increase the reaction rate by increasing the number of collisions between reactant particles?
Concentration and Temperature
Describe the differences between the 2 types of reactions: endothermic and exothermic.
Give examples for each type of reaction.
Endo: the reactants have less energy and are more stable than the products. Energy is absorbed during his type of reacton. Ex. Decomposition of water
Exo: reactants have more energy and are less stable than the products. Energy is therefore released during the reaction. Ex. combustion of methane.
What type of reaction is this?
CH4 + O2 --> CO2 + 2H2O
What is the name of this reactant CH4?
Combustion; methane
AgNO3 + Na2CrO4 -->
2AgNO3 + Na2CrO4 -> Ag2CrO4 + 2NaNO3
Au(NO3)3 + KI -->
Au(NO3)3 + 3KI --> AuI3 + 3KNO3
What is a catalyst. How does it impact the rate of a chemical reaction?:
A substance that is added to a reaction to help to accelerate the rate of the reaction by decreasing the amount of activation energy needed for the reaction to occur. A catalyst in NOT consumed during a chemical reaction.
Example KI in the decomposition of H2O2
Draw an energy diagram for an exothermic reation. Make sure to include the following:
reactants, products, ac tivated complex, activation energy, delta H, axis titles

Identify, complete and balance this chemical reaction:
BaCl2 + Pb(NO3)2 -->
Double replacement
BaCl2 + Pb(NO3)2 --> Ba(NO3)2 + PbCl2
C3H7OH + O2 -->
2C3H7OH + 9O2 -> 6CO2 + 8H2O
Sn(OH)2 + HClO3 -->
Sn(OH)2 + 2HClO3 -> Sn(ClO3)2 + 2H2O
Describe how surface area impacts the rate of a chemical reaction.
In which condition is surface area not an applicable factor to reaction rate?
When you increase the surface area of solid reactant, you increase the area available for reactions to occur. This increases the overall rate of the reaction. ie. cutting a solid into smaller pieces, or making it into a powder.
Liquids and gases
Draw an energy diagram for an endothermic reaction.