Releases energy
Exothermic
True or False:
During a phase change, the kinetic energy changes.
False, potential energy changes
When a buzzer is ringing, is it in a high energy or low energy state?
high energy
Fill in the blank: Heat transfers from ____________ to ____________.
hot, cold
Convert 56 degrees Celsius to Fahrenheit
132.8 degrees fahrenheit
Absorbs Energy
Endothermic
What phase change is a gas to a solid?
deposition
How could you get the lights in our classroom to return to a low energy state?
Turn them off.
List the following substances in order from lowest specific heat capacity to highest: water, sand, towel
2. Towel
3. Water
As temperature increases, what happens to the particles in the substance?
They move faster.
When two or more phases are present at the same time
Equilibrium

At what time (in minutes) does the liquid first appear?
2 minutes

What is increasing between 6.5 minutes and 20 minutes?
Potential Energy

Which substance is easier to heat?
Porcelain
Definition of Temperature
Measurement of average kinetic energy of particles
Definition of Triple point
When all three phases (solid, liquid, gas) are in equilibrium
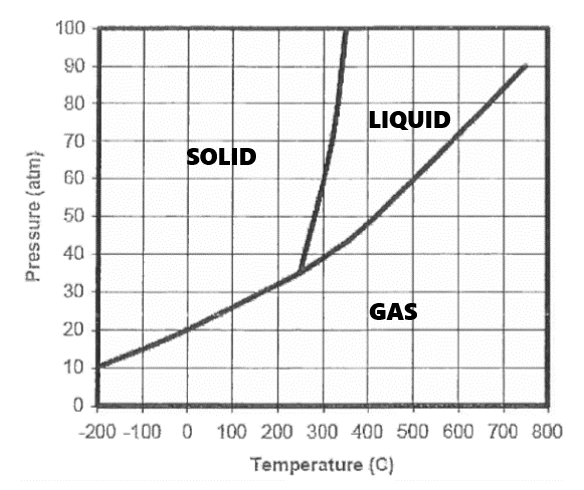
If the substance starts at 45 atm and 700°C, then heat is continually removed to decrease the temperature, it will __________ from a _____ to a _______, then __________ from a _______ to a _____.
condense, gas, liquid, freeze, liquid, solid
 In this heating curve, when is kinetic energy decreasing?
In this heating curve, when is kinetic energy decreasing?
Never
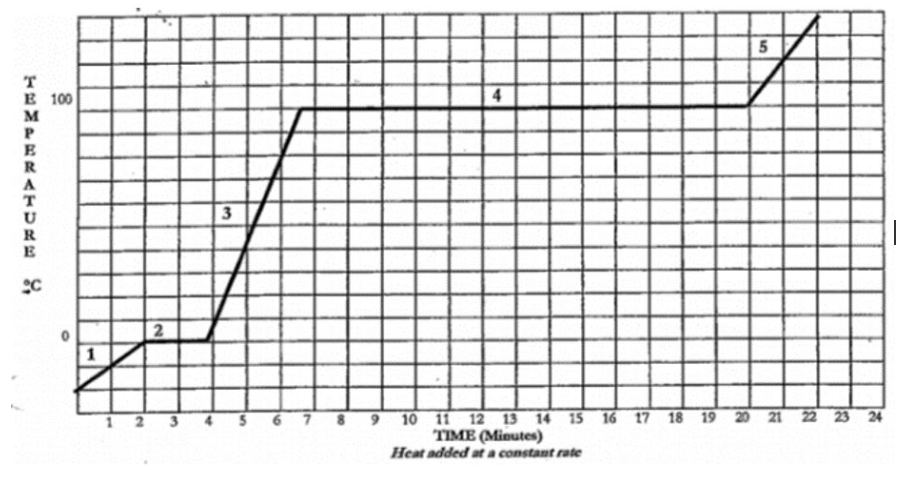
According to this graph, from 0 to 22 minutes, what is constantly increasing?
Heat
As a space heater is ran, what happens to the particles in the air around it?
they vibrate faster/kinetic energy increases
"How much heat is needed to change a substance's temperature by one degree" is called
Specific Heat Capacity

What temperature AND pressure is this compound in equilibrium between a solid and a liquid?
Answers may vary.
List, in order, 3 main types of energy used in a battery powered flashlight
chemical, electric, light
 If the same size samples of these substances were left out in the sun for the same amount of time, which material would be lower in temperature?
If the same size samples of these substances were left out in the sun for the same amount of time, which material would be lower in temperature?
Wax
 In this heating curve for water, at what temperature (in Fahrenheit) is there equilibrium between a liquid and a gas?
In this heating curve for water, at what temperature (in Fahrenheit) is there equilibrium between a liquid and a gas?
212 degrees fahrenheit