What is a fuel?
A substance which burns to release energy
What is the molecular formula for methane?
CH4
What is the molecular formula for butene?
C2H4
Which 2 elements are found in hydrocarbons?
Hydrogen & Carbon
Name a biofuel
Any from:
biodiesel, ethanol, biogas, cooking oil
What are the 3 requirements for fire?
fuel, oxygen and heat
What is the general formula for the Alkanes?
CnH2n+2
What is the general formula for the Alkenes?
CnH2n
What is a use for kerosene?
jet fuel
Hydrogen is not thought of as a biofuel because it's not made from plants but it is a clean fuel because when it burns what is the only product?
Clue:
Hydrogen + oxygen --> ?
water
Give an example of a Fossil Fuel and give an approximate number of years that they took to form
Examples - coal, oil, natural gas.
300 million years
Name this alkane
Pentane
Name this alkene 
Hexene
As you go down the tower, the boiling point ___?____, as does the number of carbons in each molecule increases
increases
Ethanol is a biofuel made from corn or sugar cane. What process is needed to convert the sugar to ethanol?
Fermentation (followed by distillation)
What does 'Exothermic' mean?
A reaction that releases heat.
Are alkanes saturated or unsaturated?
Saturated
Are alkenes saturated or unsaturated?
Unsaturated
Where are the least viscous hydrocarbons found in the distillation tower?
at the top
How is biodiesel made?
By filtering used cooking oil
Fuel + __?___ ---> ___?___ + water
Fuel + Oxygen ---> Carbon Dioxide + water
Name this molecule. 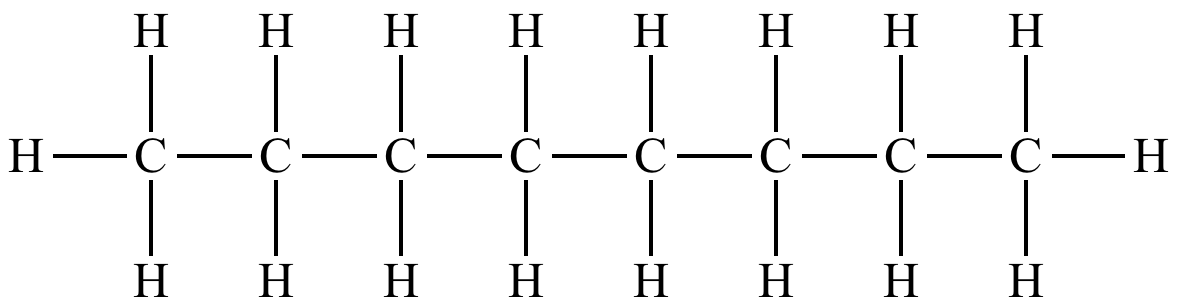
Octane
Why do the Alkenes begin at ethene and not methene?
The Alkenes contain a Carbon-to-carbon double bond. Ethene has the minimum 2 Carbon atoms. Therefore, Methene does not exist.
Give a use for bitumen
tarring road
Biofuels are renewable. What does the term "renewable" mean?
It means they are easily replaceable (within a lifetime)
What is the chemical test for oxygen?
it relights a glowing splint
hydrogen is not a hydrocarbon because it is a very flammable fuel. What is the chemical test for hydrogen?
it burns with a pop
How can we test if a hydrocarbon is unsaturated?
Add bromine and the bromine will decolourise
Give a use for naphtha
making plastic and other chemicals
Name two greenhouse gases that we are trying to cut down on (by burning biofuels instead of fossil fuels)
carbon dioxide
methane