What is temperature related to? (HINT: We learned about this type of energy in Unit 1)
Average Kinetic Energy
Siria receives the following assignments scores in Science:
98 , 98 , 98 , 98
What is her average in Science?
98. Anytime you have the same number, the average is always the same. You do not have to waste time calculating it.
What is the main difference between bar graphs and line graphs?
Bar graphs can show different things at one point in time, but line graphs show the same thing over time.
How do molecules transfer kinetic energy and change the temperatures of other molecules?
Collisions
When working with the energy cube model, where each cube represents one unit of kinetic energy, what is the correct way to determine the THERMAL ENERGY of an object?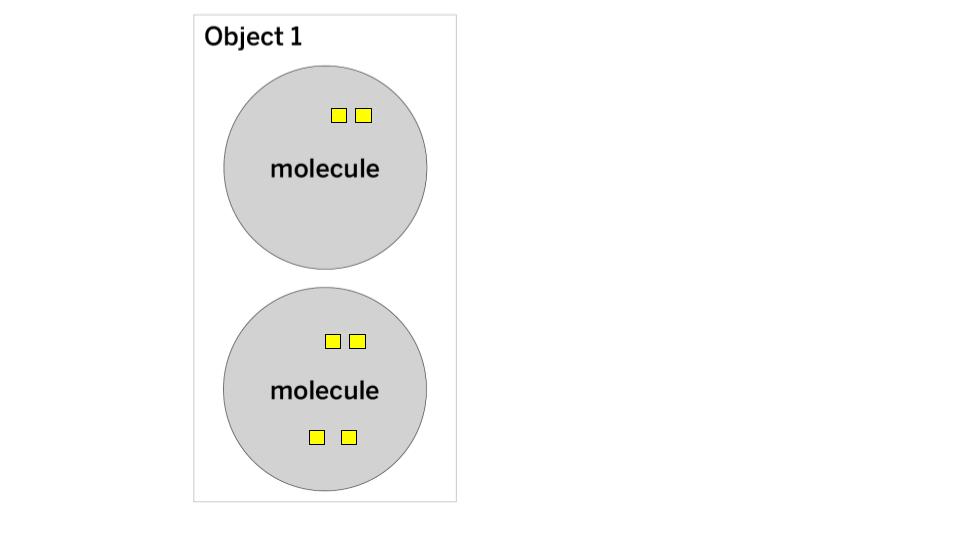
Add up all of the energy cubes.
What is the main difference between TEMPERATURE and THERMAL ENERGY?
Thermal Energy is total kinetic energy of the molecules of a thing and temperature is the average kinetic energy of the molecules of a thing.
Yesterday was a hot day. Describe the molecules of the air.
The molecules were moving fast.
Explain the three steps of calculating an average.

I want to compare the total number of students wearing BK Studio gear on BK Studio Gear Day for each class. Should I use a bar graph or line graph? Why?
We would use a bar graph to show the multiple class totals at one point in time.
The starting temperatures of two samples are:
SAMPLE A: 30℃
SAMPLE B: 20℃
After putting them in contact, what would be their ending temperature?
25℃
The ending temperature is always halfway between, or the exact middle of the two starting temperatures. This exact middle can also be calculated by finding the average.
When working with the energy cube model, where each cube represents one unit of kinetic energy, what is the correct way to determine the TEMPERATURE of an object?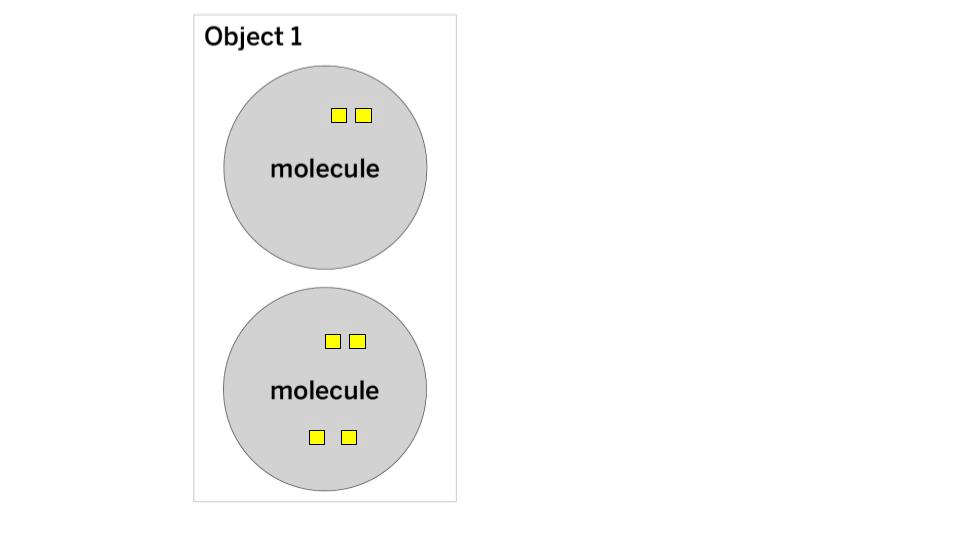
Add up all of the energy cubes, count the number of molecules, and then divide the total of all the energy cubes by the number of molecules.
Is it possible for a cold pizza pie (8 slices) to have more thermal energy than a hot slice of pizza? Why?

Yes- because even if the cold pie has less average kinetic energy, it still can have more total kinetic energy than the hot slice since it has more molecules.
Which sample is hotter?
BONUS QUESTION: How do you know?
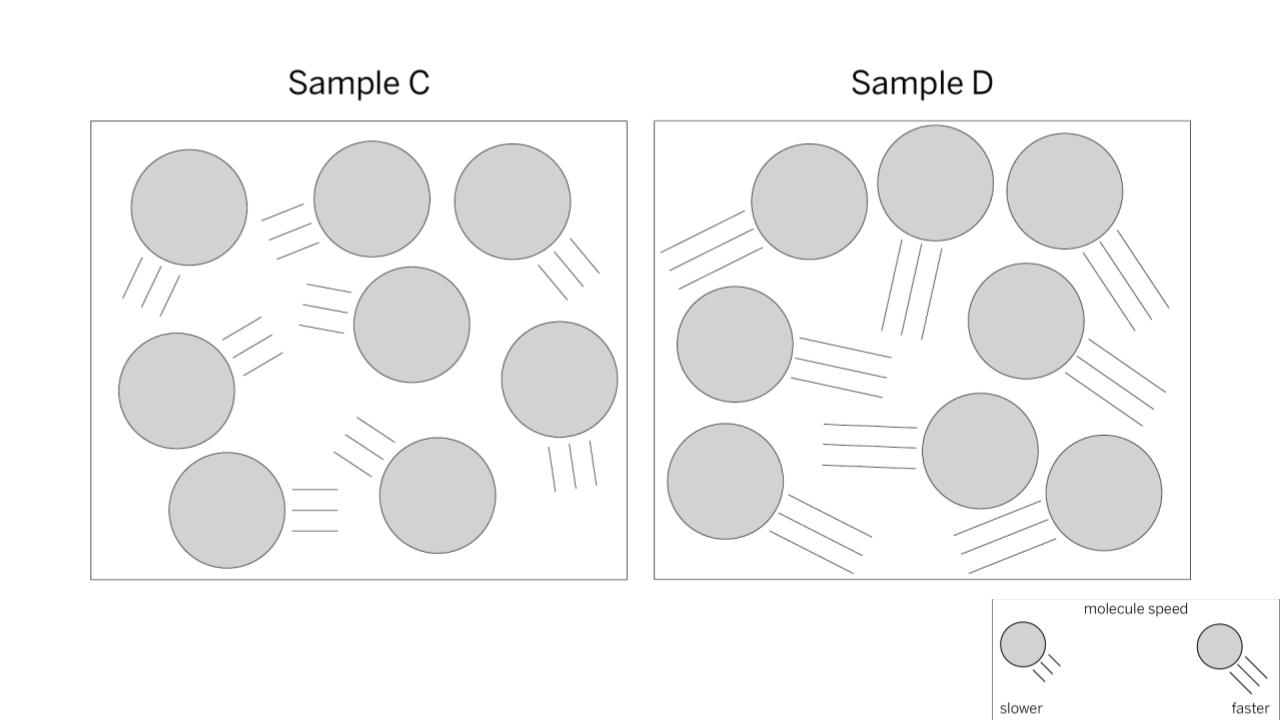
Sample D. Hot temperatures means that molecules are moving faster. The model shows faster moving molecules with longer tails. Sample D shows molecules with longer tails.
Maylin observes the following amount of squirrels come to the BK Studio grass each day.
Mon- 2
Tues - 3
Wed - 3
Thur - 5
Fri - 2
On average, how many squirrels come to the BK Studio grass?
STEP 1:
2+3+3+5+2 = 15
STEP 2: 5 days
STEP 3: 15 / 5 days = 3 squirrels per day
I want to record how many birds visited the BK Studio lawn from September to June. Should I use a bar graph or line graph? Why?
We would use a line graph to show one thing (the amount of birds) over the course of time.
TRUE OR FALSE: When two samples come in contact and reach the same temperature, their molecules stop moving.
FALSE: They do not stop moving, they just move at the same speed. If they stopped moving, this would be absolute zero.
In this energy cube model, each cube represents one unit of kinetic energy. What is the THERMAL ENERGY and TEMPERATURE of Object 1?
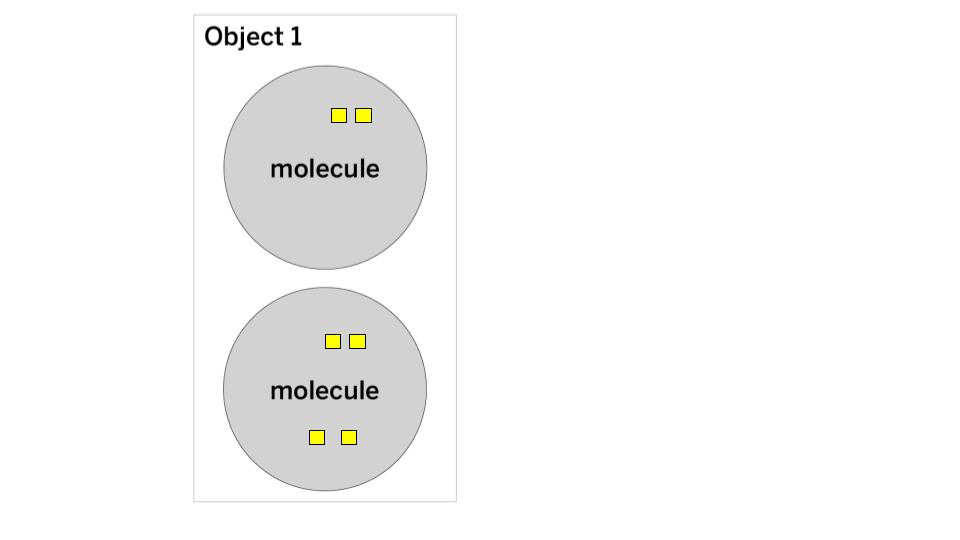
The thermal energy is 6 (2+4 = 6).
The temperature is 3 (Step 1: add 2+4 = 6, Step 2: count the number of molecules = 2, Step 3: Divide 6/2 = 3)
Which has more thermal energy- a small, hot cup of coffee OR a large iceberg? Why?
The iceberg has more thermal energy because it has so many more molecules than the cup of coffee and thermal energy is total kinetic energy. Even if the kinetic energy for each molecule of the iceberg is low, there are still so many more molecules that the total kinetic energy is much higher.
Which images correctly display a hot and cold sample?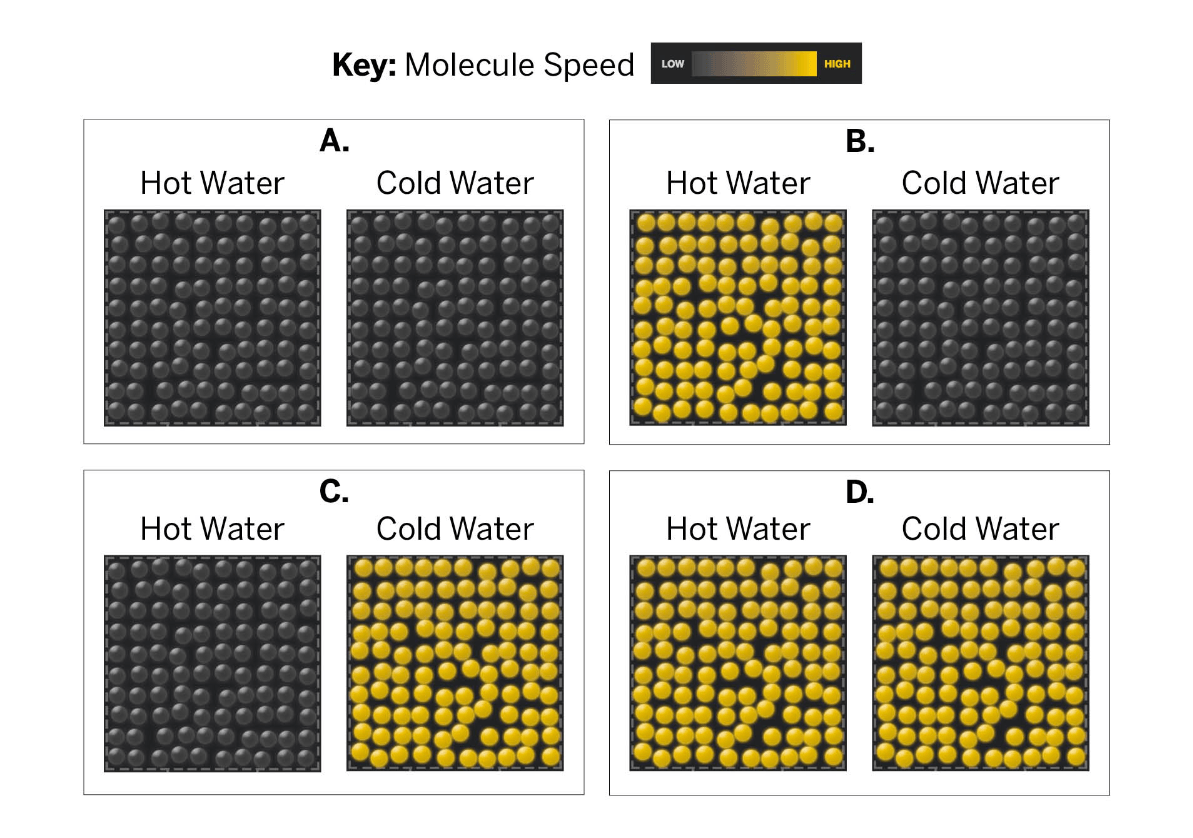
Letter B.
Faster moving molecules are shown in bright yellow, similar to our simulation. Hot temperatures means molecules are moving faster. Letter B is the only one to show fast moving molecules (yellow) for the hot water and slow moving molecules for the cold water.
Which sample is hotter? You must explain to get the points.
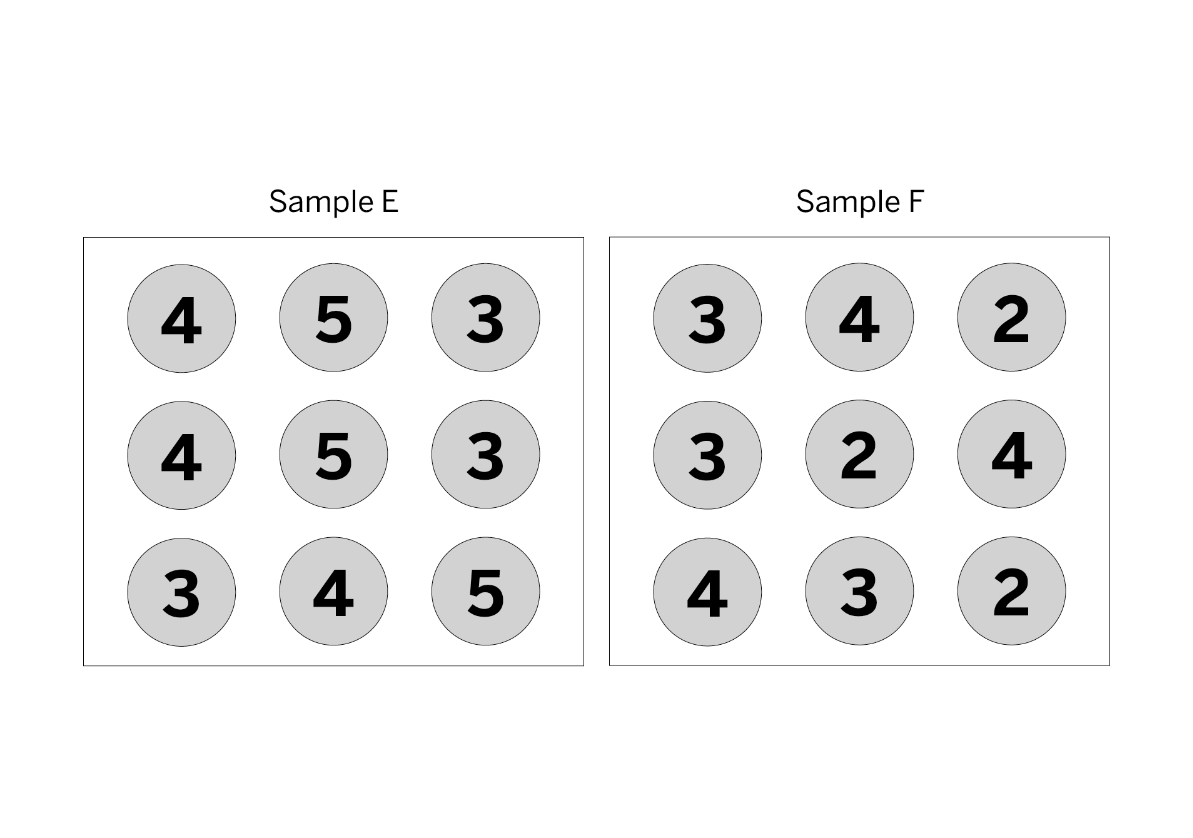
Sample E because it has a higher average kinetic energy.
You would find the average kinetic energy of both samples. The one with the higher average kinetic energy has a higher temperature.
Sample E
STEP 1: 4+5+3+4+5+3+3+4+5= 36
STEP 2: 9 molecules
STEP 3: 36 / 9 molecules = Average KE of 4
Sample F
STEP 1: 3+4+2+3+2+4+4+3+2= 27
STEP 2: 9 molecules
STEP 3: 27 / 9 molecules = Average KE of 3
4 is more than 3
The line graph below was created after analyzing a simulation of two samples coming into contact with each other. Describe what is probably happening at 13 seconds according to this line graph.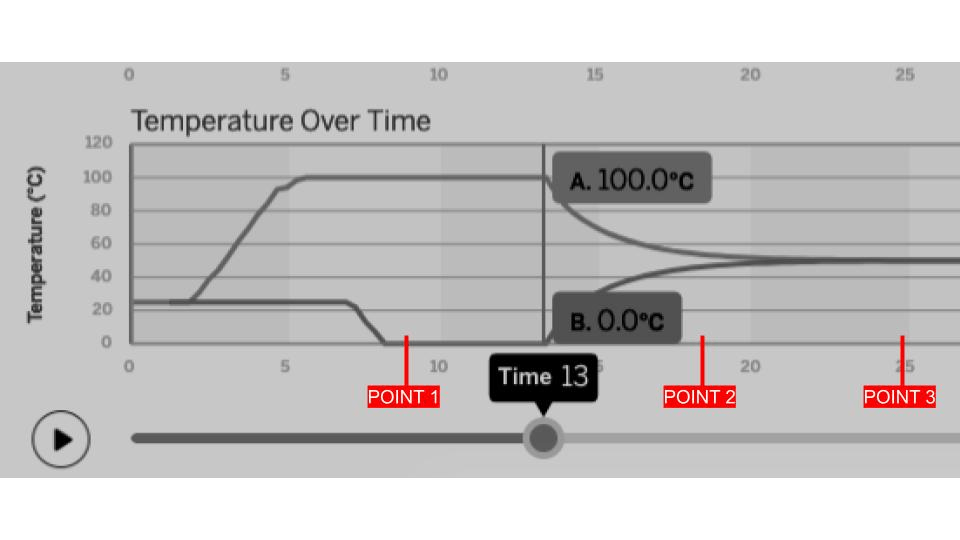
The two samples came in contact with each other and started transferring energy.
Do molecules ever stop moving?
If they did, it would be called absolute zero, but so far, it has never happened, even though scientists in labs have come close.
In this energy cube model, each cube represents one unit of kinetic energy. What is the THERMAL ENERGY and TEMPERATURE of each object?

The thermal energy of Object 1 is 4 and the thermal energy of Object 2 is 8.
The temperature of Object 1 is 2 and the thermal energy of Object 2 is 4.
Each circle represents a molecule and the number in the circle represents the kinetic energy of the molecule. Which object has more THERMAL ENERGY? Why?
Object A has more thermal energy because it has more molecules than Object B (all with the same kinetic energy as the molecules in Object B).
Explain why food dye would spread slower in a cold water sample than a hot water sample.
USE THESES WORDS:
KINETIC ENERGY
MOLECULE
TEMPERATURE
The molecules of the water with a higher temperature move faster, or, have more kinetic energy. Therefore, they would spread the food dye around faster.
Which sample is hotter? You must explain to get the points.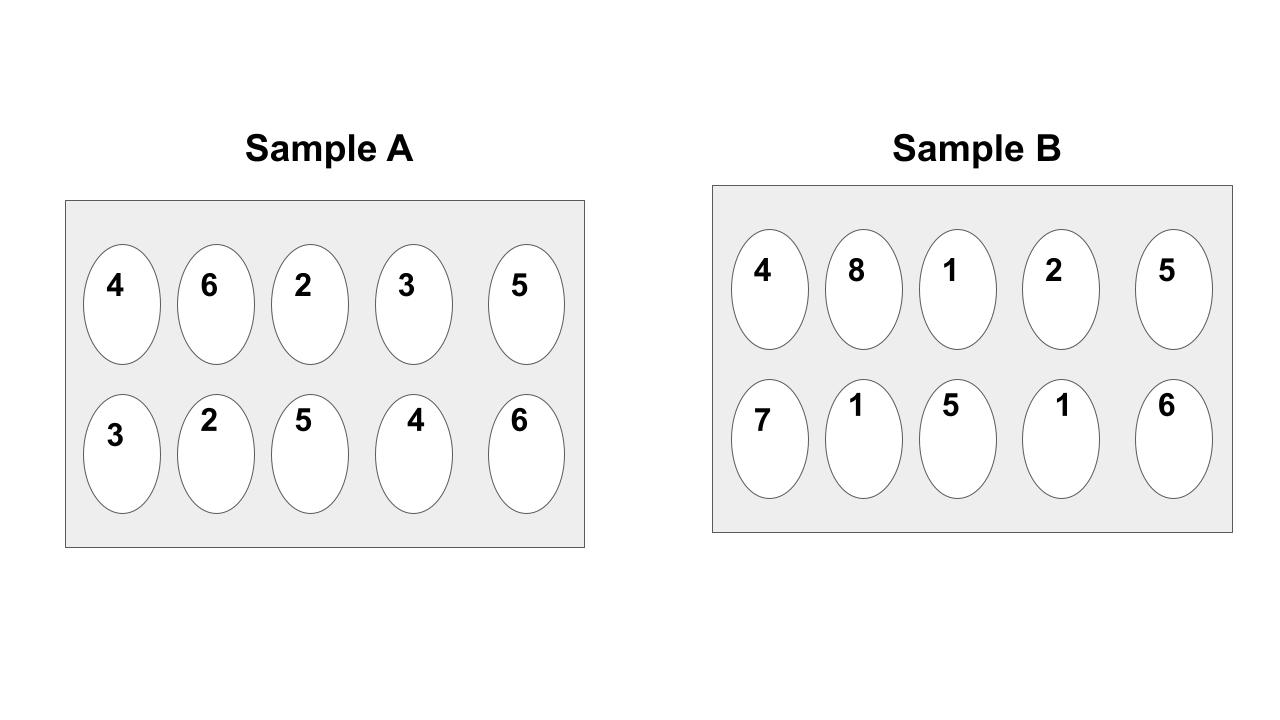
They are both the same temperature because they have the same average kinetic energy.
The line graph below was created after analyzing a simulation of two samples coming into contact with each other. Which point shows when both samples reached EQUILIBRIUM and why?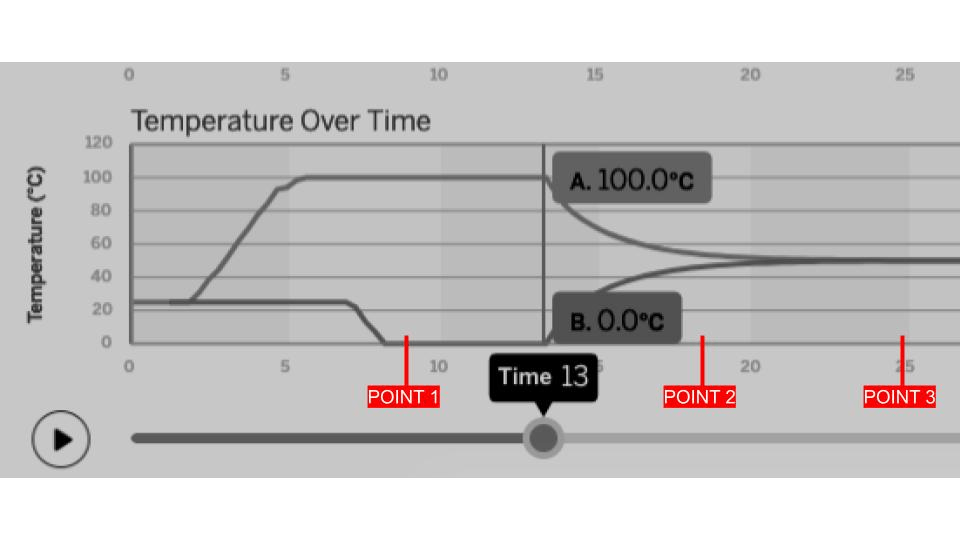
Point 3 because both samples have the same TEMPERATURE.
Draw what is happening when the heater heats up the molecules of air.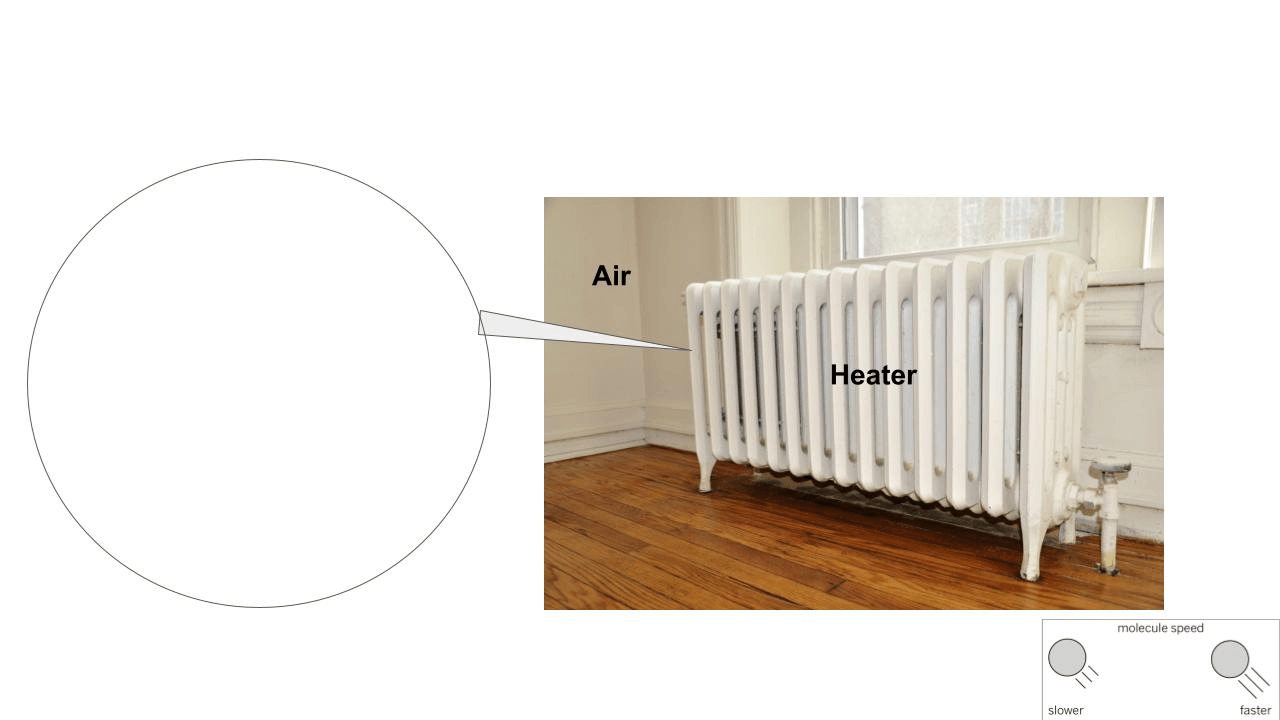
The zoom in on the image should show faster molecules colliding into slower molecules.
In this energy cube model, each cube represents one unit of kinetic energy. If both objects are put in contact with each other, what will the final EQUILIBRIUM TEMPERATURE of both objects be and why?
The equilibrium temperature of Object 1 and Object 2 will both be 3 because each molecule in Object 2 will transfer one energy cube to the molecules of Object 1.
Each circle represents a molecule and the number in the circle represents the kinetic energy of the molecule. Which object has a higher TEMPERATURE? Why?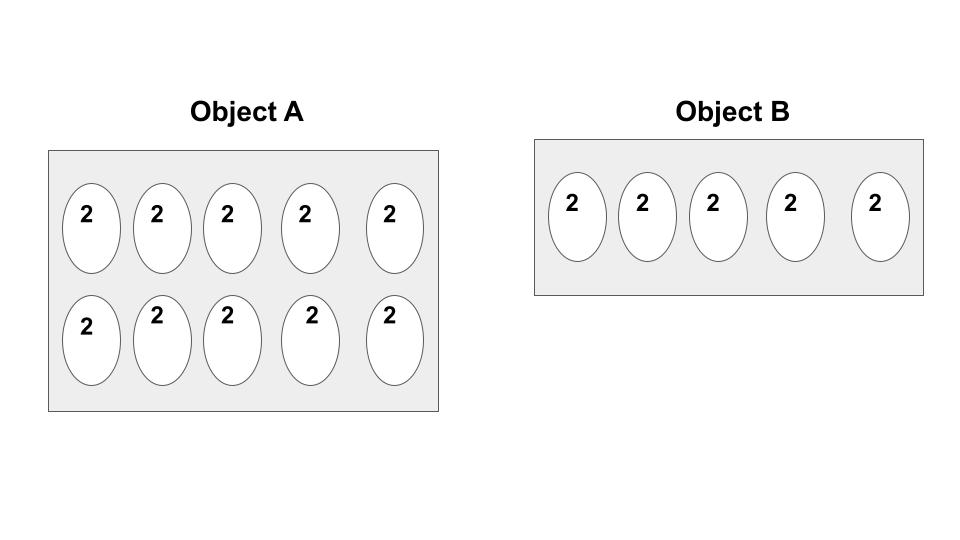
Both objects have the same temperature because temperature is the average of the kinetic energy and the average for both of the objects is 2.