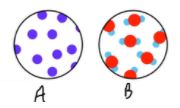
Which state of matter would be considered the most dense?
Solid
A change from one substance to another is what kind of change?
Physical or Chemical
Chemical
If phosphorus became an ion by gaining an electron, what would be it's charge?
-1
What are the vertical columns and the horizontal rows called on the periodic table?
Vertical = Groups
Horizontal = Periods
When an atom loses or gains an ___________, an ion is formed.
Electron
Identify the substance A in the picture:
compound, mixture, or element
Element
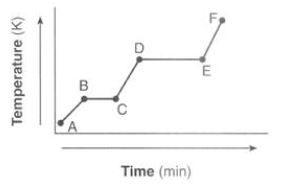
What state is occurring from D to E?
Boiling
Neutrons and _________ have almost the same mass.
Protons
How many electron rings are found in Chlorine (Cl)?
3
Moving left to right across a row of the periodic table, which of the following values increases by exactly one from one element to another?
Atomic Number
Cereal and Milk is an example of a ____________ mixture.
Heterogeneous
The heat of vaporization is the amount of energy required to change a substance from a ________ to a ________.
Liquid to a Gas
If an atom has 18 protons and 20 neutrons, which element is it?
Argon
The energy required to remove an electron.
Ionization Energy
How many valence electrons does Boron have?
3
The temperature at which a liquid reaches enough pressure to break away and become a gas is....
Boiling Point
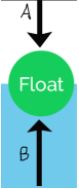
Which arrow represents the buoyant force?
B
If an atom of tin has a mass number of 118, and an atomic number of 50, how many neutrons are in its nucleus?
68
The tendency of an element to attract electrons.
Electronegativity
The _____________ is the weighted average of an elements isotopes.
Atomic Mass
a Mixture where different materials remain distinct.
Heterogenous Mixture
measuring the resistance in the friction of "flow" is called what?
Viscosity
Which of the following is unique to each individual element?
Protons
What element on the periodic table has the smallest atomic radius?
Helium
Oxygen-18 and Oxygen-17 are considered what?
Isotopes