Who described atoms as small spheres that could not be divided into anything smaller?
a. Bohr
b. Dalton
c. Rutherford
d. Thomson
b. Dalton
The smallest particle of an element
An atom
Describe the part of the atom marked "B”?

Electrons are located here, they also have negative charged.
True or False... Isotopes can be the same element.
True
What does 14 stand for in the isotope carbon-14?
Mass number
Metals are located where on the periodic table?
To the left and the middle of the staircase
Elements On the Periodic Table are placed in __________________ or families in which they have similar chemical properties.
Groups
Two atoms with the same number of protons but different numbers of neutrons.
Isotope
The first model of the atom was developed through
a. scientific experimentation and data analysis with water vapor and gases.
b. observation of the light emitted by elements when they are heated.
c. thinking about the smallest particles of matter without experimenting.
d. analyzing the effects of a magnet on the path of a cathode ray.
c. thinking about the smallest particles of matter without experimenting.
This is the very small, dense center of an atom.
The Nucleus
Students are completing a table about a particular subatomic particle that helps make up an atom.

What best explains how they should complete the table?
a. with “Inside the nucleus,” because the particle is a proton
b. with “Inside the nucleus,” because the particle is a neutron
c. with “Outside the nucleus,” because the particle is a proton
d. with “Outside the nucleus,” because the particle is an electro
b. with “Inside the nucleus,” because the particle is a neutron
Isotopes have the same protons, but these are different.
Neutrons
What is the hyphen notation of this isotope?
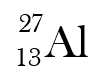
Aluminum-27
Nonmetals are located where on the periodic table?
To the right of the stair case
This number tells you the number of protons in the nucleus of every atom of an element.
Atomic Number
The model shows a calcium atom. How many electrons are in the first level?

2
What did Rutherford’s model of the atom include that Thomson’s model did not have?
nucleus and electron cloud
These two particles make up the nucleus of an atom.
Protons and neutrons
How does an atom change if all of its electrons are removed?
a. The atom’s volume increases.
b. The atom’s mass increases.
c. The atom becomes positively charged.
d. The atom becomes negatively charged.
c. The atom becomes positively charged.
This makes Carbon-12 and Carbon-14 isotopes. What is the same about these two isotopes?
Same element, same atomic number, and same number of protons
Free-Points
Free-Points
These are the rows of the periodic table and the properties of the elements in these rows change as you move from left to right.
Periods
This is how zirconium appears in the periodic table.
Round to the nearest whole number, how many electrons are in an atom of zirconium?

40
How many valence electrons is depicted in the diagram?

2
What did Bohr’s model of the atom include that Rutherford’s model did not have?
energy levels/clouds
What are the three particles found in an atom?
Protons, Neutrons, and electrons
Explain why the overall charge on an atom is zero?
The positive charge of the protons in the nucleus equals the negative charge in the electron cloud.
Free-Points
Free-Points
How would you write out a hyphen notation for magnesium atom that has 12 protons and 15 neutrons
Magnesium-27
Metalloids are located where on the periodic table?
On the staircase in between the metals and nonmetals
One of the first Periodic Tables was constructed by Dmitri Mendeleev, He arranged the elements by increasing ______________, ______________, _____________, and ____________.
Atomic Mass, Valence Electrons, Reactivity, and Physical State.
Determine which Bohr model is depicted in the image below.

Oxygen
The plum pudding model of the atom states that
a. each atom has an overall negative charge.
b. each atom has a dense, positively charged center.
c. an atom is a solid, indivisible sphere that makes up all matter.
d. an atom is made up of electrons in a sea of positive charges.
d. an atom is made up of electrons in a sea of positive charges.
What two particles in an atom are equal in number.
electrons and protons
What value is closest to the mass of the atom? Also, include the unit.

10 amu
True or False... Atom A has 13 protons and 14 neutrons. Atom B has 14 protons and 13 neutrons.
Atom A and Atom B are isotopes.
False. Isotopes have the same number of protons.
The hyphen notation name for an isotope with 46 electrons and 61 neutrons.
palladium-107
Sulfur (atomic number 16) belongs in which group number?
group 6

Letter D is the mass number of an element that tells you the average mass of the isotopes of the element. It also tells you the number of ____________ and ____________ in the nucleus of the atom
protons and neutrons
Determine which Bohr model is depicted in the image below.
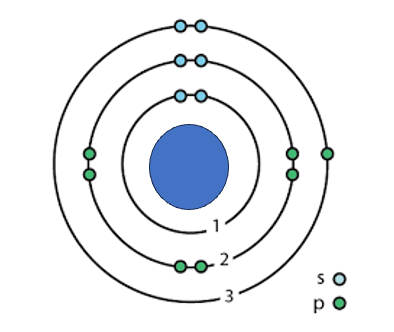
Aluminum