If the system is at equilibrium Q <=> K?
=

At what time is the system at equilibrium? 0, 20, 21, or 60
What is 60 seconds?
This stress only affects equilibrium reactions with at least one gas
What is a pressure change?
Write the Ksp expression for the following dissociation: Mg(OH)2 (s)
Ksp= [Mg2+] [OH-]2
If K>1, what side of the equation is favored? Reactants, Products, or neither?
What are Products?
If the system is shifting to the left Q __ K
What is >
 Which system shows equilibrium? 1-2- or 3
Which system shows equilibrium? 1-2- or 3
What is reaction system 2?
Fe3+ + SCN- <------> FeSCN2+ If I added Fe3+ to the system the reaction would favor....
What are the products?
1.8x10-11= 4s3
1.8x10-11/4 = s3
(1.8x10-11/4)1/3= s
This is the only stress that can change the Kc value
What is temperature?
A + 2B --> AB2 Kc= 100
If a vessel has [A]= .1M [B]=.2 and AB2 is .5, is the system at equilibrium or will it shift to the left or right?
What is shift to the LEFT?
.5/[(.2^2)x.1]= 125 100<125; K<Q
This addition of this will speed up BOTH the rate of the forward AND reverse reactions, so does not get included in the equilibrium expression?
What is a catalyst?
N2O4 <---> 2NO2 ^H= +58 kJ In order to favor the products the temperature would need to...
What is increase?
What are the units of molar solubility in a Ksp equation?
mol/L
When flipping an equation, the Kc
a. is multiplied by -1
b. is held constant
c. is raised to the -1 power
C. raising to the -1 is the reciprocal
A + B <---> C + D
A 32 L flask has 16 mol of A and B and 32 moles of C, and D. If Keq is 5.5, determine which way the reaction is running
To the right!
K> Q 5.5> 4 1x1/(.5x.5)=4
These two states of matter are not included in equilibrium expressions and do not affect the constant.
What are solids and liquids?
5A (g) + 9D (l) <---> 6E (aq) + 3G (g) If we increase the volume, which way will the reaction shift?
What is shift to the LEFT?
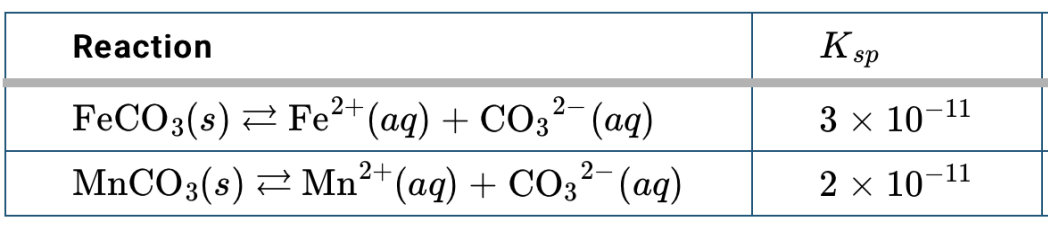
What solid will have more ions in solutions?
FeCO3
H2 + F2 <---> 2HF Initially, 10 mol H2 and 6 mol F2 are in an evacuated 2L flask. At eq, 5 mol HF are present. Write the initial row for the ICE table

Star= 5
? = 3
??= 2.5 NEED TO USE CONCENTRATION
mol/L

D. Q= 1,851 > K
K<Q shifts to the left, so products decrease
4A (g) + 2B (g) <---> 3C (g) What is the equilibrium expression, Kp?
MUST USE () for P
Kp = (Pc)3 / (PB)2(PA)4
If the temperature of an exothermic reaction increases, what happens the Keq?
K<1, so you are making the K value smaller/decreases
AgBr has a Ksp of 5x10-13. If a student adds NaBr to a test tube with AgBr, what happens to the molar solubility?
What is decreased?
Remember Ksp is a constant, only changed by temperature
N2 + 3H2 <---> 2NH3
Initially, 12 mol NH3 are put in an evacuated 2L flask. At equilibrium, the concentration of N2 is 1 M. What is the Keq?
0 0 12/2=6
+x +3x -2x
1 3 4 4^2/(1x3^3)
16/27= .59