What charge does an electron have?
-1
What is the unit of current?
AMPERE
What type of ions do metals form?
CATIONS (POSITIVE)
What is the name of a salt made from hydrochloric acid?
Chloride
What happens to reaction rate when temperature increases?
Increases
What happens when two objects with the same charge are brought close together?
They repel
State Ohm’s Law.
Current is proportional to potential difference.
Voltage = Current x Resistance
What happens to a metal atom when it forms an ion?
It loses electrons
Magnesium is added to hydrochloric acid. What are the products of this reaction?
Magnesium chloride and hydrogen gas
Explain how increasing concentration affects the frequency of collisions between particles.
More collision occur because reactants are more likely to meet one another.
Which subatomic particle is responsible for static electricity?
The electron
A resistor has a resistance of 5 Ω and a current of 2 A flows through it. What is the voltage?
10 Volts
A metal reacts with water. What does this tell you about its position in the reactivity series?
Very high due to high reactivity
A student reacts calcium carbonate with nitric acid. List the three products of this reaction.
calcium nitrate, water and carbon dioxide.
Three experiments are carried out:
A: Cold, dilute acid + iron powder
B: Cold, dilute acid + iron lump
C: Hot, dilute acid + iron powder
Place the experiments in order of increasing rate of reaction.
B < A < C
A PVC rod is rubbed with a wool cloth.
Explain which way electrons move during this process and state the charge on:
the PVC rod
the wool cloth
Electrons move from wool to PVC
PVC = negative
Wool = positive
A 12 V battery is connected to two resistors in parallel: 6 Ω and 4 Ω
Calculate:
1. the current in each branch
2. the total current from the battery
6Ω branch = 12V/6Ω = 2A
4Ω branch = 12V/4Ω = 3A
Total current = 5A
A student adds iron metal to a solution of zinc sulfate.
Will a reaction occur?
Give a reason based on the reactivity series.
No. Iron is below zinc in the reactivty series.
A student mixes sodium carbonate solution with calcium chloride solution.
A double displacement reaction occurs.
What are the products of the reaction?
Calcium carbonate and sodium chloride
Name the shaded region on an exothermic reaction energy diagram and the substances that reduce it. (hint anagram ACTS SALTY)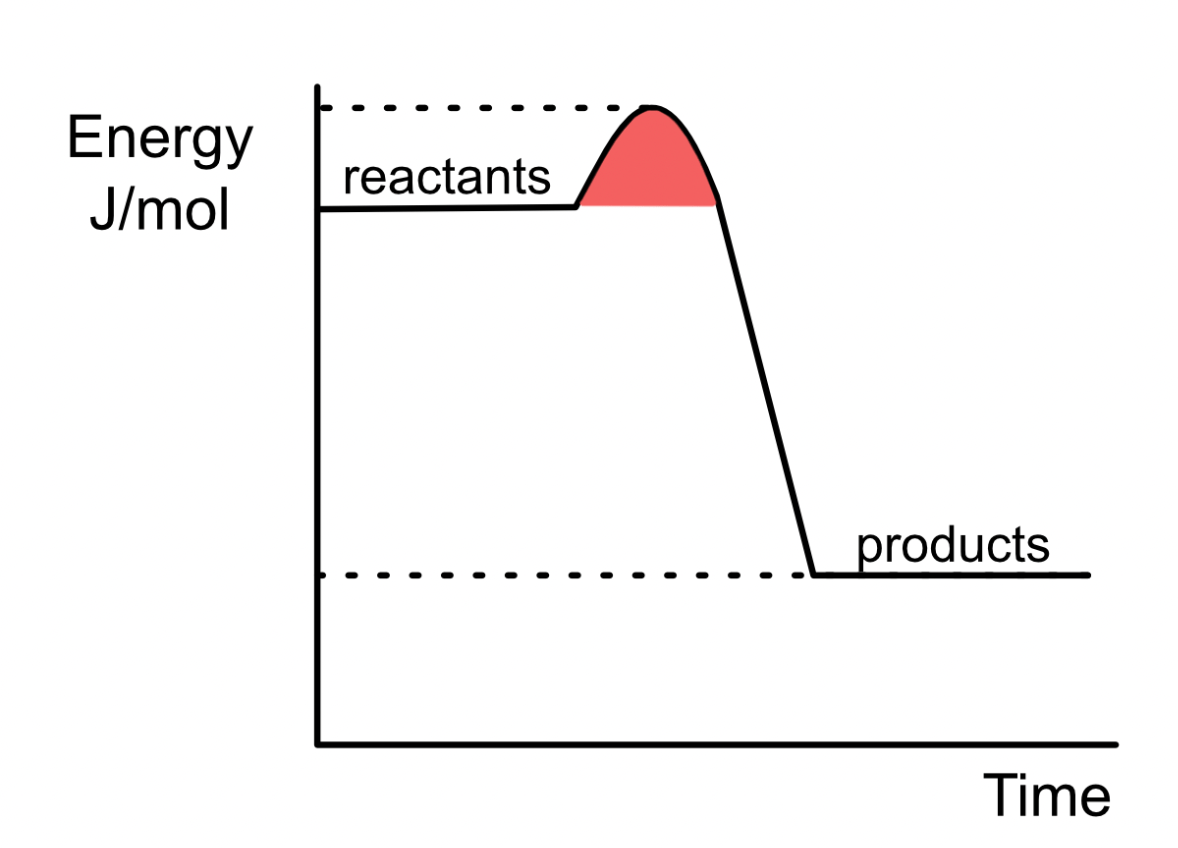
ACTIVATION ENERGY AND CATALYSTS
A negatively charged PVC rod is brought close to several small, neutral strips of paper.
The strips move towards the rod without touching it.
What is the name of the process causing this attraction?
INDUCTION
A 12 V battery is connected to two resistors in parallel.
The total current is 8 A. One branch has a resistance of 3 Ω.
Calculate:
the current in the 3 Ω branch
the current in the second branch
the resistance of the second resistor

12V/3Ω = 4 A
∴ current in other branch = 8 - 4 = 4 A
∴ x = 12V/4A = 3Ω
Carbon is used to extract some metals from their ores.
Which metal cannot be displaced by carbon?
- iron
- zinc
- aluminium
Aluminium
A student reacts sodium hydroxide solution with hydrochloric acid until neutralisation is complete.
Which salt is formed?
Which gas is released?
Sodium chloride AND no gas
What name is given to the biological catalysts that reduce the activation energy of a reaction?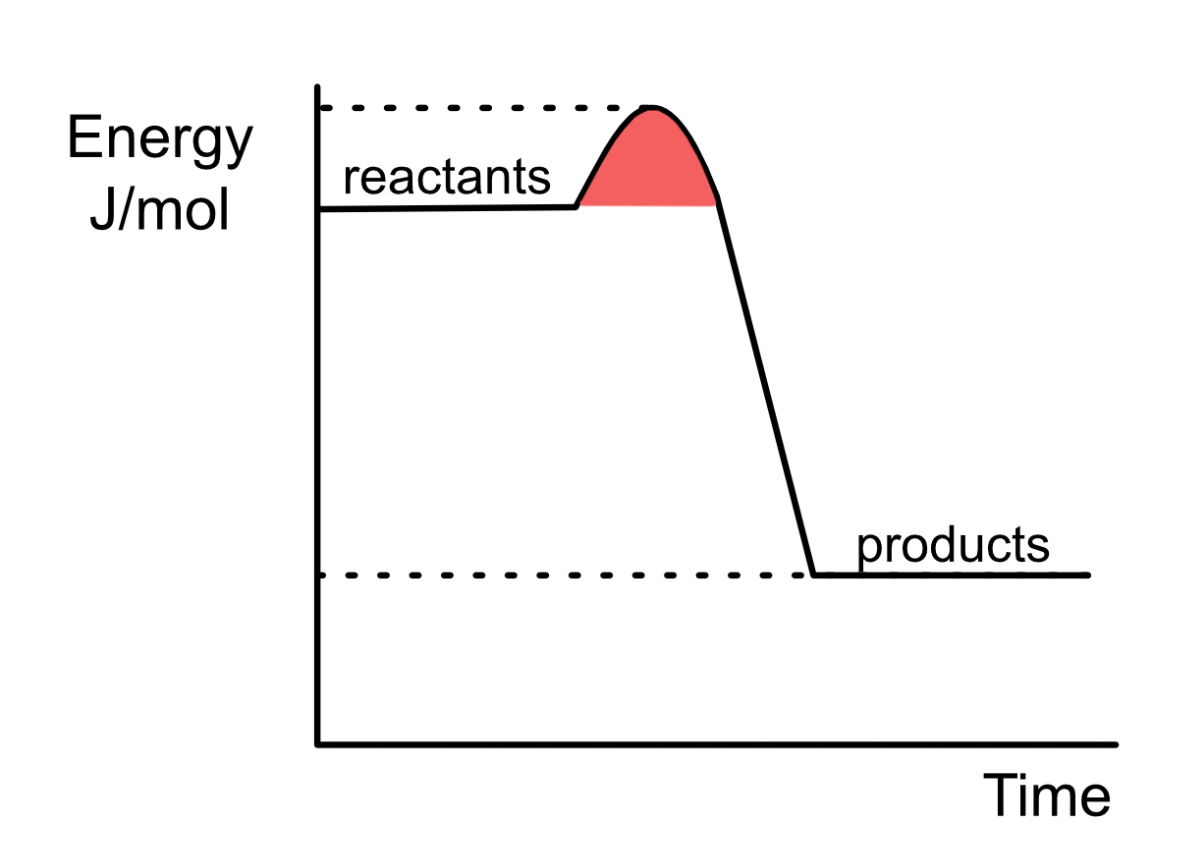
ENZYMES