What is the general formula for the Alkanes?
Protons & Neutrons
What is the Nucleus made up of?
Exothermic
What is a negative Enthalpy Change?
Temperature
What increases the rate of reaction?
KCr2O7
What is a oxidising agent used for the oxidation of Alcohols?
Dumb Bells
What is the shape of a P orbital?
293-298
What is Room Temperature in Kelvin?
2-
What is the Oxidation number of Oxygen?
Smaller Concentration
What will decrease the rate of reaction?
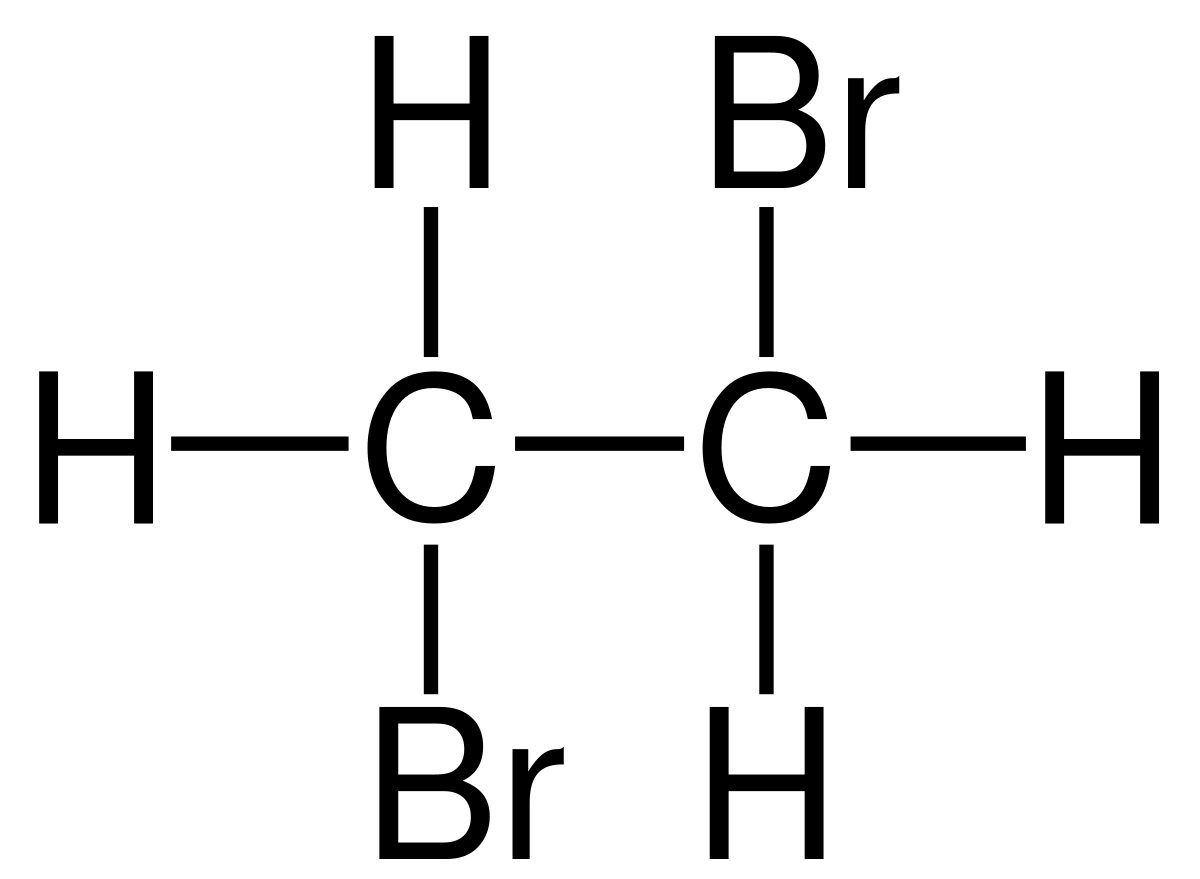
What is the display formula of Di-bromomethane?
1s2 2s2 2p6 3s2
A Born-Haber Cycle
What are used to calculate the Lattice Enthalpy of Formation of one mole of a molecule?
An element that has been oxidised.
What is Change in Concentration/Time ?
An electron pair acceptor.
What is an Electrophile?
Ar 4s2
What is the electron configuration of Calcium?
The Energy required to break one mole of a specified type of bond in a gaseous molecule.
What is Average Bond Entalpy?
Phenolphthalein.
What indicator is used in Titrations?
Catalyst
Used for Drug Testing in sport.
What can Mass Spec be used for?
104.5°
What is the bond angle of a Non-Linear compound with two lone pairs?
Specific Heat Capacity
What is the amount of energy required to raise the temperature of 1g of a substance by 1°K
Concordant
A catalyst with different physical state to the reactants
What is a heterogeneous catalyst?