To balance complex redox equations, which atoms should be balanced first?
All atoms other than oxygen and hydrogen
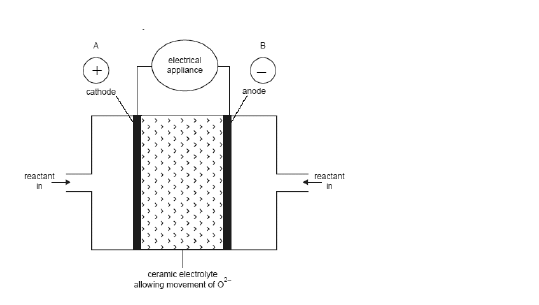
cathode
What is the energy conversion that occurs in a fuel cell?
chemical to electrical
Are the reactions that occur in galvanic cells spontaneous or non-spontaneous?
spontaneous
How much does a 1st year teacher get paid in Victoria?
How much does a 1st year teacher get paid in NSW?
$79 589
$90,177
Where on the electrochemical series would you find the strongest oxidant?
top left hand corner
What is the polarity of the anode in a galvanic cell?
negative
How do you balance a carbonate fuel cell?
Balance the C, then H normally, then balance the oxygens with CO3- on one side and add an equal amount of CO2 on the other side
How would you balance for a SOFC
Balance the oxygens using O2-
What is the spell to levitate in Harry Potter
Wingardium levi-OOOOOsa
What is the oxidation number of S in H2SO4?
+6
Explain the function of a salt bridge
provides negative and positive charged ions that can migrate into half cells to balance the charge. Without a salt bridge, a galvanic cell will cease to operate.
Hydrogen fuel cells can be used to power cars. What disadvantages are there in the use of hydrogen as a fuel?
Safety and cost of storage and transport (explosive, very light gas must be pressurised or liquefied)
Write the half equations for the acode and cathode

How many Tim Tam packets tall is Ms Tenedora?
Hint: a Tim Tam packet is 220mm long
7 tim tam packets (according to chatgpt)
Define oxidant, and what reaction does it undergo?
Hint: is it always a product or reactant?
- an oxidant (or oxidizing agent) is defined as a reactant that removes electrons from another species and gets reduced in the process
How is the potential voltage of a galvanic cell calculated?
cell potential difference = E0 of half-cell containing oxidant - E0 of half-cell containing reductant
Why are porous electrodes used in fuel cells?
Increase surface area to maximise rate of reaction
The > indicates the flow of O2-. Identify the:
-anode
- cathode
- direction of flow of e-

Name this pokemon:

Ekans
Solid Mg (s) is placed in an aqueous Al(NO3)3 solution. Describe two observations that can either be measured or seen.
- Mg(s) decrease in size/mass
-H2 gas produced
Explain how standard electrode potentials (Eo values) are obtained.
By combining half-cells with the standard hydrogen half-cell.
Explain the function of the electrolyte in a fuel cell
Allows the transfer of ions participating in the reaction from one electrode to another
Write thehalf equations for the cathode and anode
cathode: 2H+(aq)+2e–→H2(g)
anode: Zn(s)→Zn2+(aq)+2e–
In 1932, the Australian army was sent to WA with machine guns to fight a war. Who was the war against?
Emus