Ionic Bond
Covalent bonds form between these types of elements
Name the compound made from chlorine and sodium.
Sodium Chloride
What must charges of balanced ionic compounds add up to?
Zero
Draw a Lewis Dot diagram of sulfur

Type of bonding illustrated below
Ionic bonding
Atoms that have formed a covalent bond are said to do this with their electrons.
Share electrons
The balanced formula to describe a compound made between potassium and nitrogen. (ie. H2O is "H" two Oh.)
K3N
How many electrons will oxygen donate or take in an ionic bond?
It will take 2 electrons
Draw a Lewis model of O2

Bond type with 4 shared electrons
Covalent double bond
The number of electrons that participate in a double bond
4
Name of this compound:
(NH4)2O
Ammonium oxide.
A mystery element is found to have 2 valence electrons. What will the charge of it's stable ion be?
+2
Draw a Lewis diagram showing what electrons do in a bond between chlorine and sodium

Bond made when electrons are transferred
Ionic bond
draw a Lewis Dot diagram showing a molecule of N2
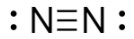
The formula of the compound made between magnesium and sulfate.
MgSO4
Write a balanced formula for a compound containing calcium and nitrogen
Draw a Lewis structure of carbon disulfide

Bond type(s) found in sodium carbonate?
ionic and covalent
4
Total numbers of protons and electrons in the polyatomic ion NH4+
Protons=11
Electrons= 10
Which type of bonding results in a network of regularly spaced charged atoms. (Network solid)
Ionic
Draw a Lewis Diagram of OH-
