A MEASURE OF THE AMOUNT OF MATTER IN AN OBJECT
UNITS IN GRAMS, KILOGRAMS
MEASURED USING TRIPLE BEAM BALANCE.
MASS
The ability of a substance to be dissolved into another substance.
SOLUBILITY
What 2 particles are found inside the nucleus of an atom?
proton
neutron
Horizontal rows on the periodic table
Periods
Pure substance that has all of the same type of atoms
They are found on the periodic table
element
Substances from 0 to 6 on the pH scale.
Sour taste.
Reacts with metals
acids
Happens when there is a re-arrangement of the molecules of a substance.
NO New Substances formed!
Can be reversed.
Ex: Changes in state of matter, shape, size, volume, mass, weight, and density.
physical change
States that matter is not created or destroyed, only rearranged or recycled.
Law of Conservation of Matter
A MEASURE OF THE SPACE AN OBJECT OCCUPIES
UNITS IN CUBIC CENTIMETERS (CM³) OR MILLILITERS (ML).
VOLUME
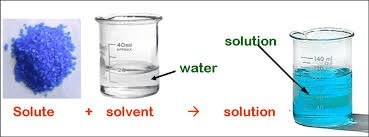
The substance that is dissolved into the solvent.
SOLUTE
Where is the electron found in an atom?
Outside the nucleus in the electron cloud
Columns on the periodic table that share the same properties.
When two or more DIFFERENT elements are combined chemically by being bonded
Turn into a new substance with completely new properties.
compound
Substances from 8 to 14 on the pH scale.
Bitter taste.
Bases
Occur when the molecules of a substance are separated or joined and changed into a new substance with new properties.
Can’t be reversed.
Ex: Bubbling and fizzing, light production, smoke, and presence of heat.
chemical change
water + CO2 = glucose + O2
photosynthesis
A COMPARISON OF AN OBJECT’S MASS TO ITS VOLUME.

DENSITY
The substance into which the solute is dissolved.
SOLVENT
Shows the number of protons in the nucleus of the atom.

Atomic number
The periodic table arranges all the elements in groups according to what?
Similar properties
When two or more substances are combined physically (in the same place), but not chemically bonded together.
mixture
Result when an acid and a base neutralize each other (cancel each other out).
Salts
The rate of a chemical reaction will _______________ when heat is added.
INCREASE
Humans & other animals breathe in oxygen and transform sugar into energy then breathe out carbon dioxide due to this process...
cellular respiration
property related to allowing electricity to pass through it easily
ELECTRICAL CONDUCTIVITY
When the solute is dissolved into the solvent.
SOLUTION
Tells us the Protons + Neutrons of an atom of that element

Atomic mass
As you move through each element on the periodic table, you add a _________.
Proton
Two or more substances that are combined PHYSICALLY, but not chemically bonded.
You get DIFFERENT amounts in each sample.
EX: Chex Mix, Italian Dressing, concrete
heterogenous mixture
Pure substances are made from one or more elements from the periodic table that are ______________ combined.
chemically
Mass cannot be created or destroyed, just changed from one form to another.
LAW OF CONSERVATION OF MASS
a complex series of processes through which carbon atoms are transferred through the ecosystem
The Carbon Cycle
property related to allowing heat energy to pass through it easily
THERMAL CONDUCTIVITY
When the solution can’t dissolve any more solute.
SATURATION POINT
In every atom, the number of these 2 particles are the same.
protons, electrons
What increases as you move through the periodic table? (Hint: there are 2 answers!)
Atomic number
Atomic mass
When one substance is dissolved throughout another substance in equal amounts.
You get the SAME amounts in each sample.
Example: Coffee, Air, Sweet Tea
homogenous mixture
Mixtures and Solutions are made of two or more substances that are __________ combined.
physically
Four siblings did chores during the weekend. Ling cooked eggs. Gabe raked leaves. Chris sewed buttons. Briana dried laundry.
WHICH IS THE CHEMICAL CHANGE?
Ling cooking eggs!
What process is occurring at X?
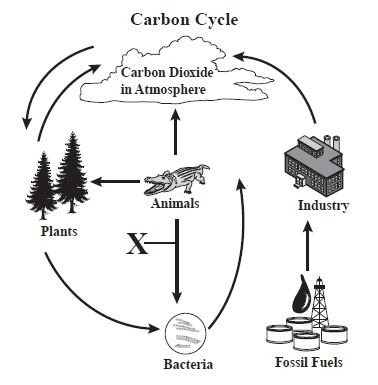
Decomposition