An intermolecular force caused by differences in electronegativity between atoms in a molecule.
What is dipole-dipole interaction?

What is hydrogen bonding, dipole-dipole, and London dispersion forces?
The name for the transition labeled 2.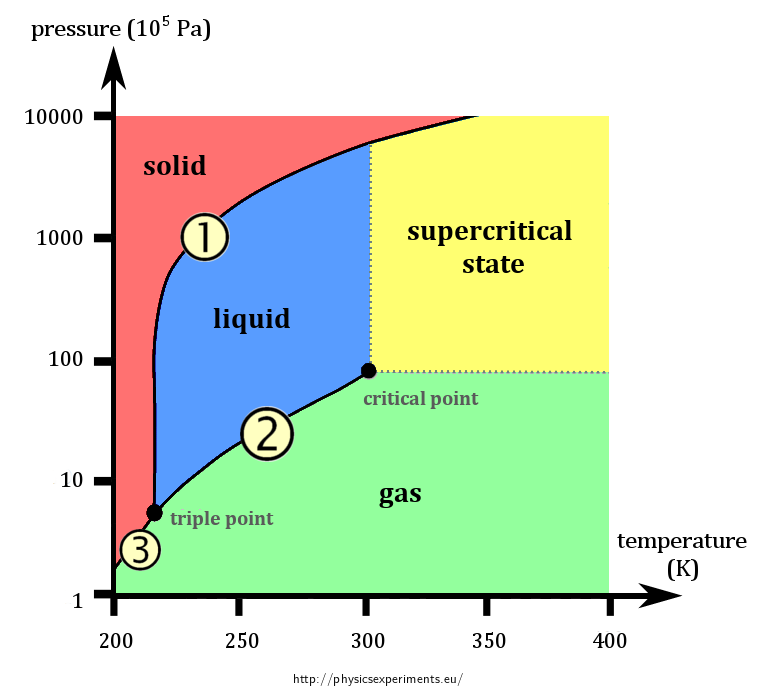
What is evaporation?
Either water (H2O) or carbon dioxide (CO2) have a higher boiling point.
What is water?
HF or HCl
HF
What is F, O, and N?
What force(s) do molecules of CH2CH2 have?
What is London dispersion forces?
The specific pressure and temperature where all three states of matter of a substance exist in equilibrium.
What is triple point?
CH3OCH2CH3 or (CH3)2CHCH3
CH3OCH2CH3
(CH3)2SO or H2O
H2O
An intermolecular force caused by a short-lived, spontaneous dipole moment.
What is London dispersion force?
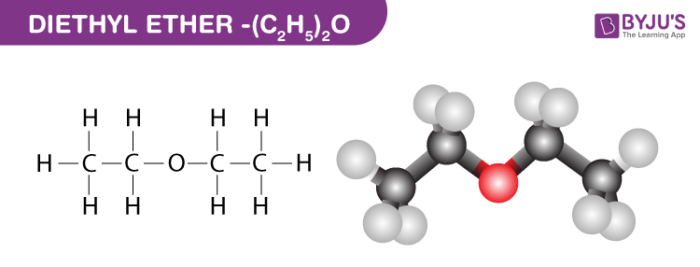 All of the intermolecular forces that diethyl ether can participate in.
All of the intermolecular forces that diethyl ether can participate in.
What are dipole-dipole interactions, and London dispersion forces?
Changing from a solid to a gas.
What is sublimation?
CH3OCH2CH3 or CH3CH2CH2OH
CH3CH2CH2OH
CF4 or (CH3)2CHCH2CH3
(CH3)2CHCH2CH3
Rank the intermolecular forces from weakest to strongest.
What is London dispersion forces, dipole-dipole interactions, hydrogen bonding?
All of the intermolecular interactions experienced in a mixture of ammonia (NH3) and water (H2O).
What are hydrogen bonding, dipole-dipole interactions, and London dispersion forces.
How is vapor pressure impacted by temperature?
As temperature increase, vapor pressure increases
CH2F2 or CH2Cl2
CH2F2
CH3CH2SH or CH3CH2NH2
CH3CH2NH2
A very uncommon type of intermolecular force that involves a polar molecule interacting with a non-polar molecule.
What is induced dipole-dipole?

What is dipole-dipole and London dispersion forces?
What happens when the applied pressure of the environment is equal to the vapor pressure of a compound?
The substance boils, it is at its boiling point.
Arrange the following molecules in increasing boiling point:
H2O, Xe, H2S, MgCl2, N2
What is N2, Xe, H2S, H2O, MgCl2
C4H9OH or C2H5OH
C4H9OH